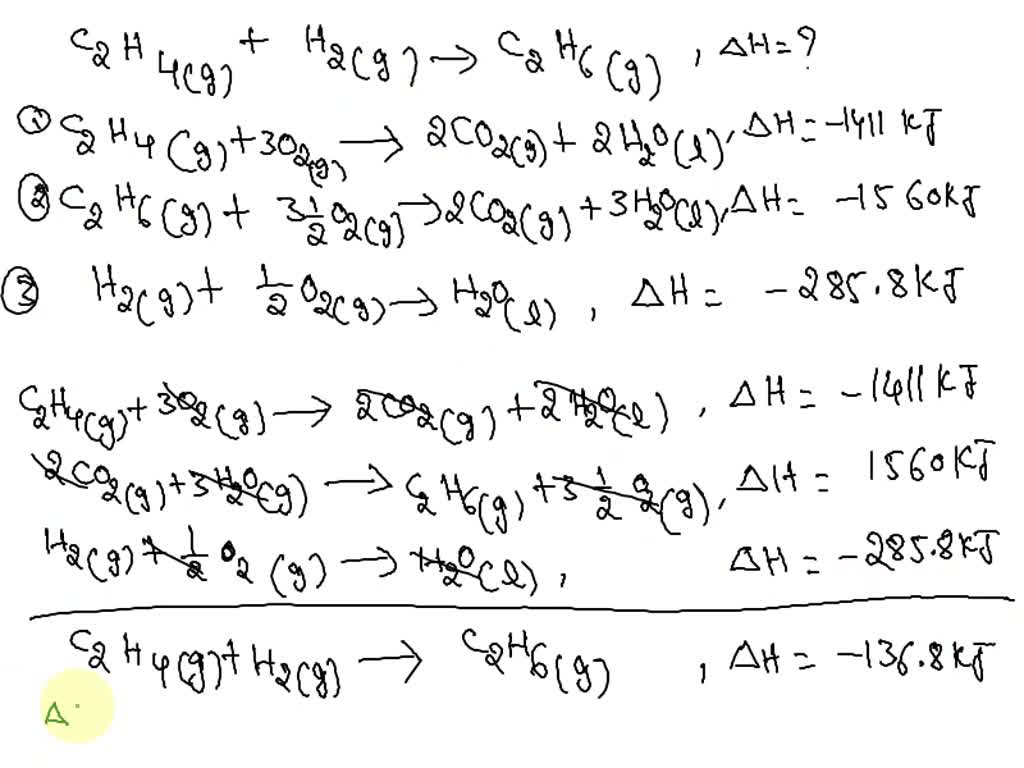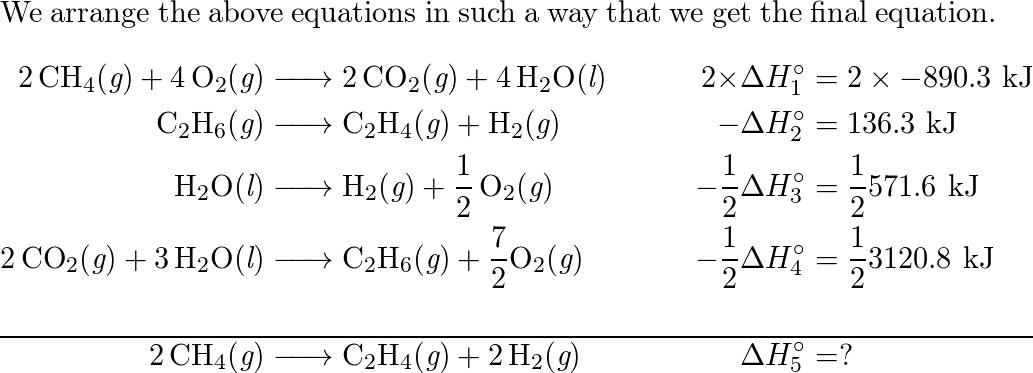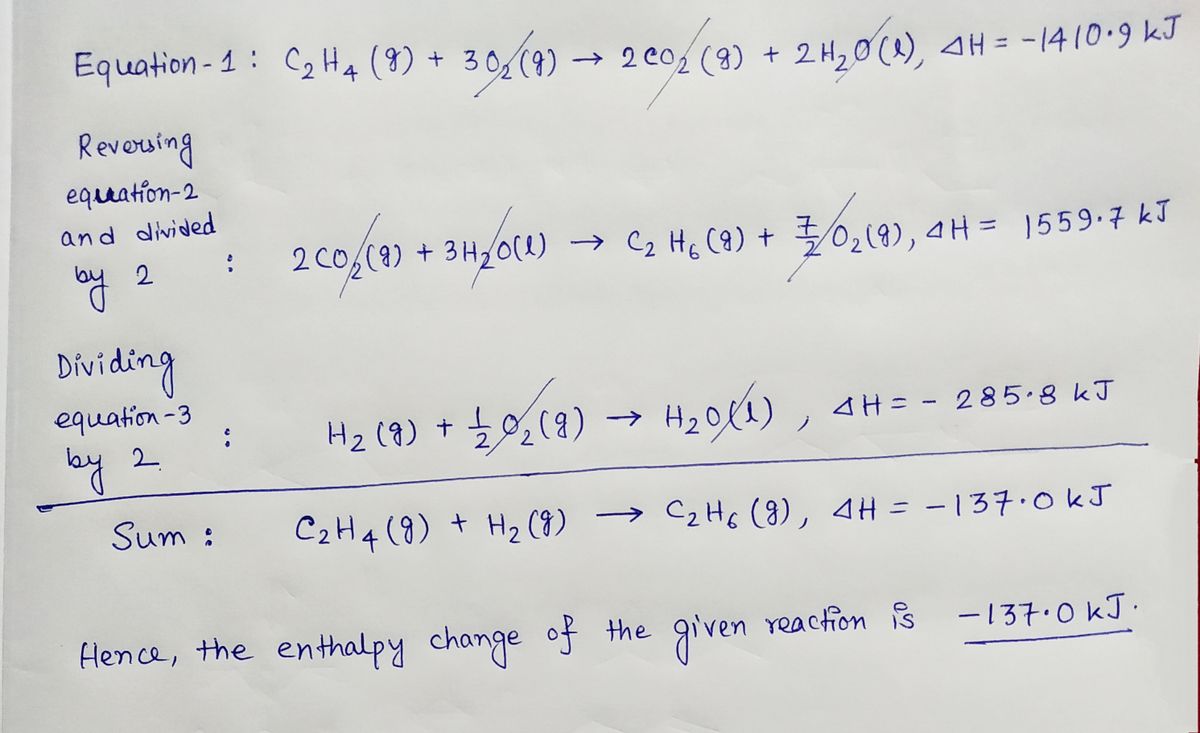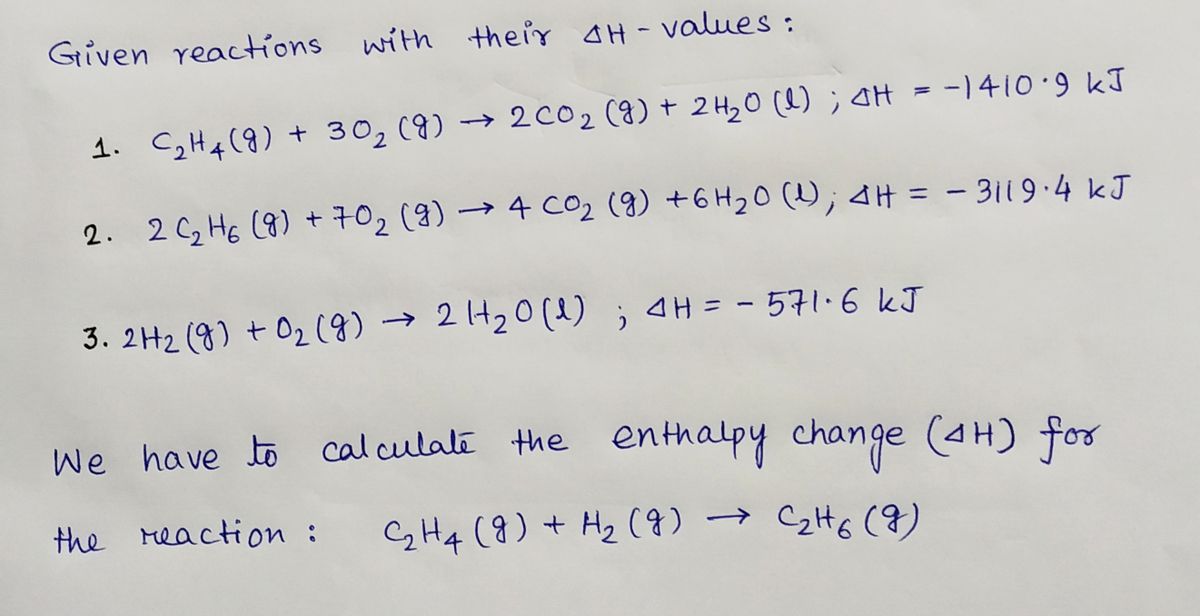Answered Calculate the enthalpy change for the bartleby
Delta g of hot sale c2h4
Share.
Visit »
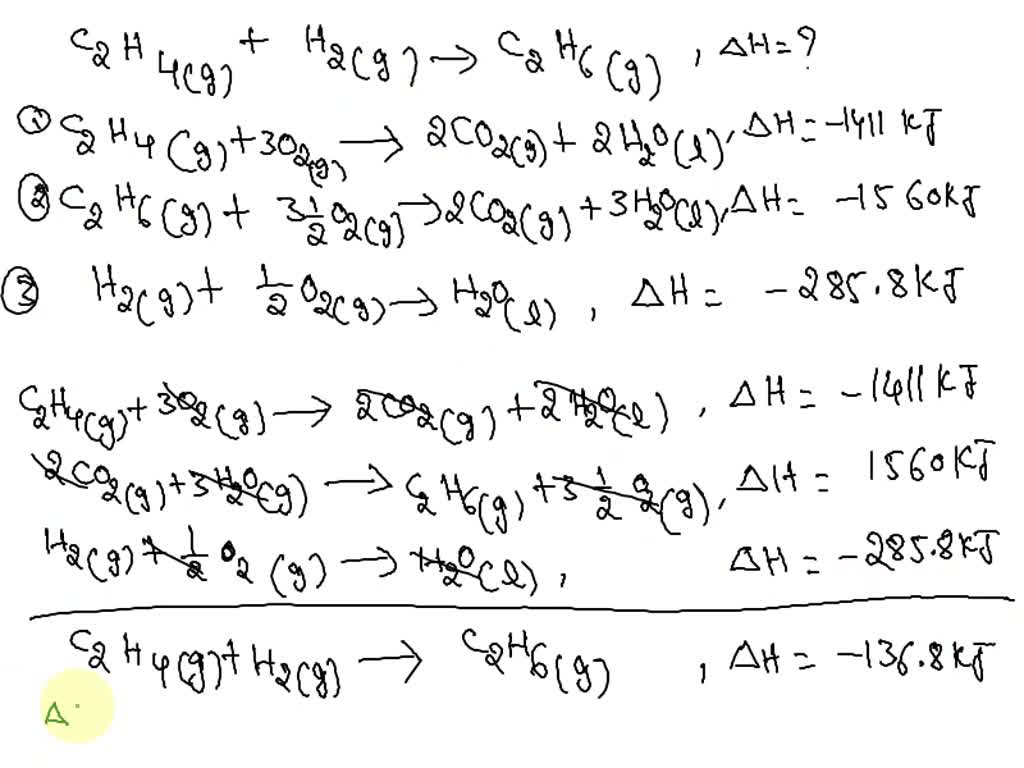
SOLVED Calculate DH for the reaction C2H4 g H2 g C2H6 
24. Delta Hf C2H4 12.5 kcal Heat of atomization of c 171 kcal 
Solved Choose the reaction that is described by the delta Chegg 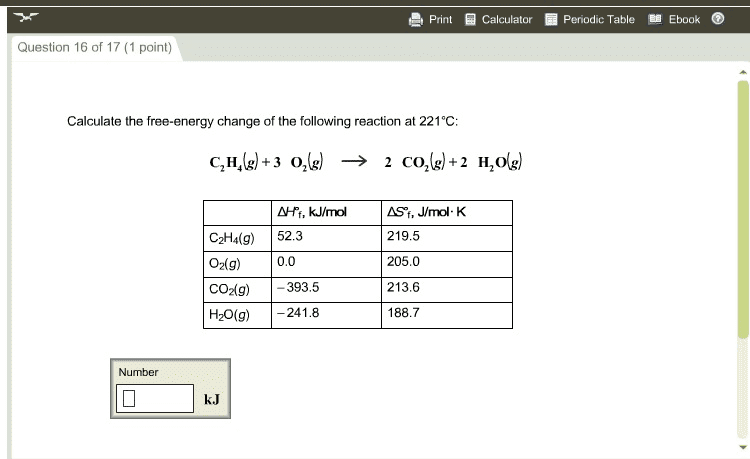
OneClass Calculate the free energy change Calculate the free 
ASSIGNED READINGS 
Be sure to answer all parts. Calculate the heats of combustion for 
Solved Consider the reaction C2H4 g H2 g C2H6 g where 
ntif enthalpies of formation for c2h4 g co2 g and h2o l at 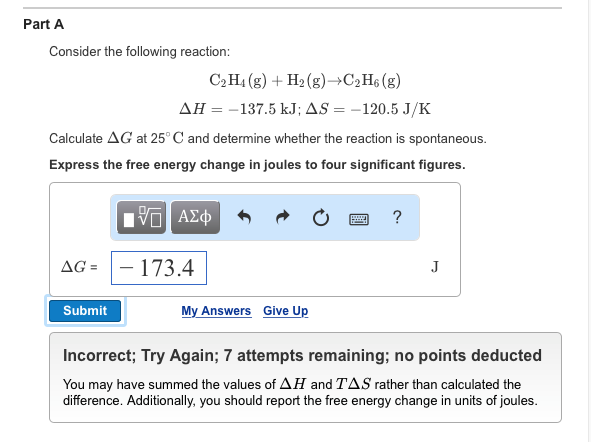
Solved Part A Consider the following reaction C2H4 g H2 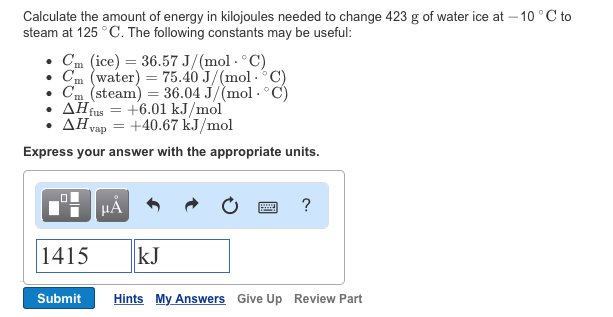
Solved Part A Consider the following reaction C2H4 g H2 
C2H6 C2H4 H2. If the enthalpy change for this reaction is 137 
SOLVED Calculate the standard entropy rxn of the 
Use bond energies to confirm that the complete combustion of 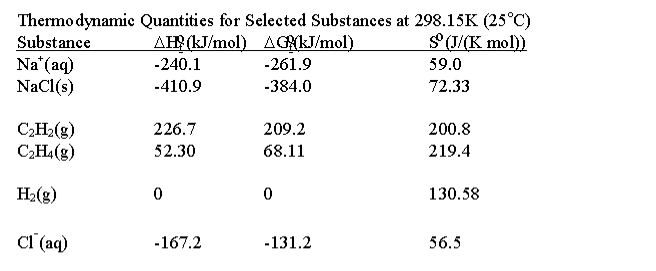
Another Thermo Question Socratic 
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g 
Solved For the reaction C2H6 g rightarrow C2H4 g H2 g 
Solved Given the standard enthalpy changes for the following 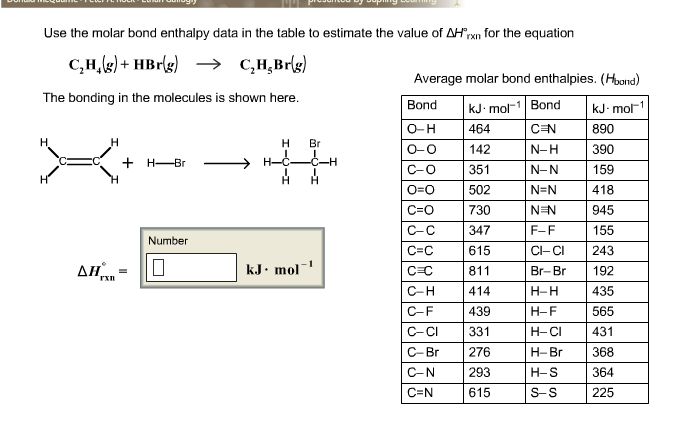
Solved Use the molar bond enthalpy data in the table to Chegg 
Find Change in Free Energy of Reaction 
Calculate the value of Delta U the following reaction C 2 H 4 g 
For the reaction C 2H 4 g H 2 g rarr C 2H 6 g K p 3.356 xx 10 17 Calculate Delta G for the reaction at 25 C 
Be sure to answer all parts. Calculate the heats of combustion for 
Consider the reaction I2 g Cl2 g 2 ICl g K p 81.9 at 
Calculate the heat of reaction 25 C the reaction C2H4 g H2 g 
Get Answer Consider the reaction C2H4g H2O g CH3CH2OHe 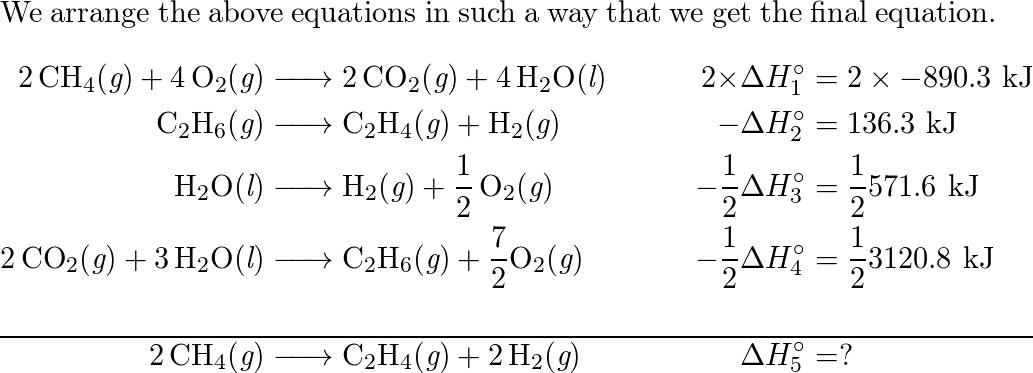
We can use Hess s law to calculate enthalpy changes that can Quizlet 
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol 
Solved Calculate the enthalpy change for the reaction Chegg 
18. Calculate the enthalpy of hydrogenation of C2H2 g to C2H4 g 
If Delta f H circ C 2 H 4 and Delta f 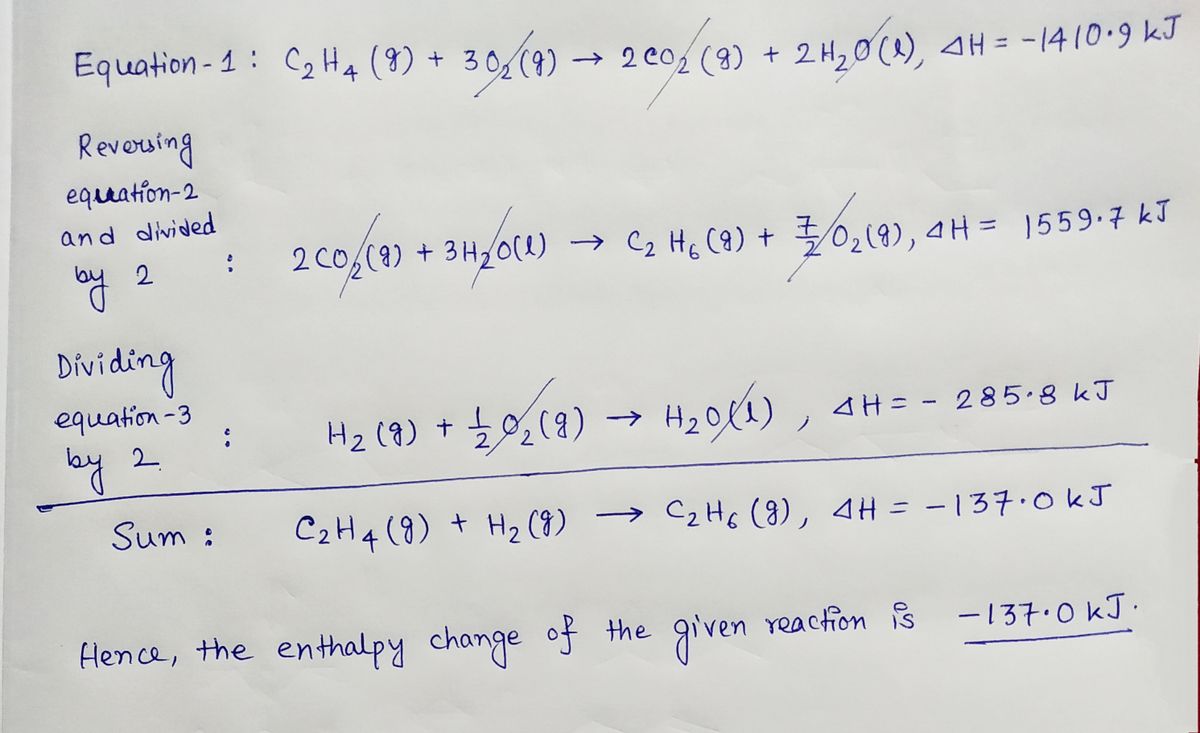
Answered Calculate the enthalpy change for the bartleby 
Standard Gibbs free energy of formation at 298 K expressed in 
Solved Values given for S 0 J mol K C2H4 219.3 H2 
7.65c Calculate the enthalpy change from bond energies C2H4 g 3O2 g 2CO2 g 2H2O g 
IN THE SOLUTION WHY IS THE UNDERLINED STEP USED H reactants H 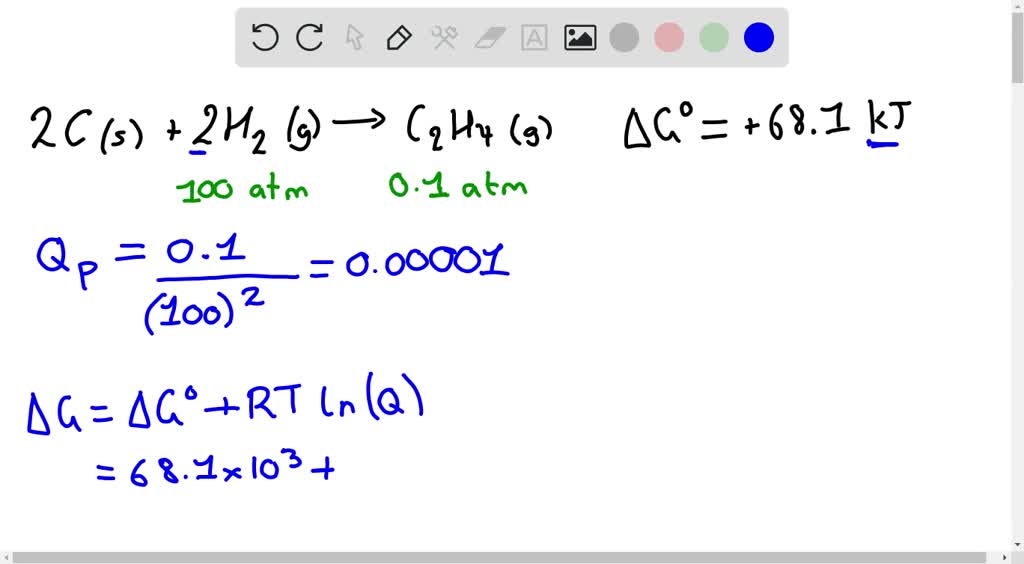
SOLVED Calculate G for the formation of ethylene C2 H4 from 
Answered The A G for the following reaction is bartleby 
Consider the following reaction C2H4 g H2 g C2H6 g delta H \\ \Delta G=-137.5 35.76\\\Delta G=-101.74\\\Delta G=Negative)
C2H4 g H2 g C2H6 g H 137.5 kJ S 120.5 J K