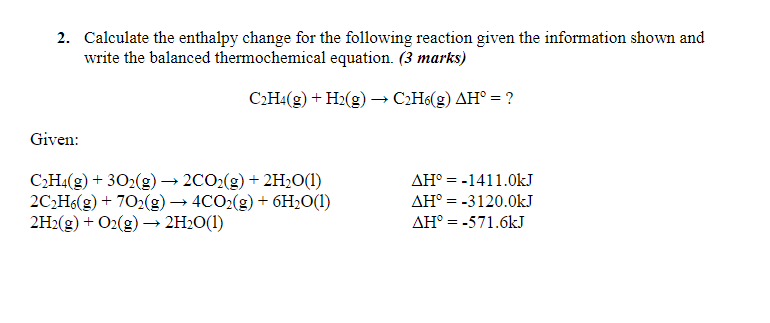Solved Given the following data 1. C2H4 g 3 O2 g 2 Chegg
C2h4 g 3o2 g hot sale 2co2 g 2h2o l enthalpy
Share. Visit »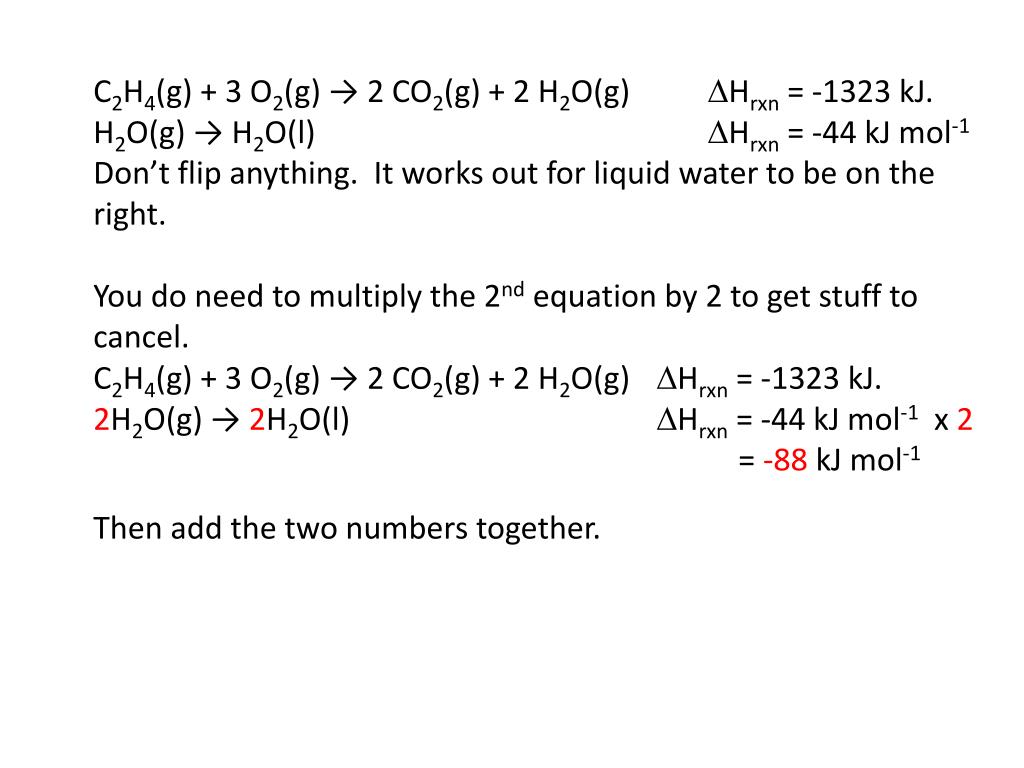
PPT Saturday Study Session 1 Theme of the Class Enthalpy H2(g) C2H6(g) using the following data..jpg)
Chapter 5 Thermochemistry. ppt download
Solved Show all your works. C2H4 g 3O2 g 2CO2 Chegg H2O(g) C2H5OH(l) 1) H2O(l) H2O(g) 2) C2H5OH(l) 3O2(g) 2CO2(g) 3H2O(l) 3) C2H4(g) 3O2(g) 2CO2(g) 2H2O(l) ΔH1= 44kJ mol-1 ΔH2= -1371kJ mol-1 ΔH3= -1409kJ mol-1.jpg)
Energetics Chemical changes caused by energy ppt download
OneClass The combustion of ethane C2H4 occurs via the reaction
Solved Calculate H for the following reaction C2H4 Chegg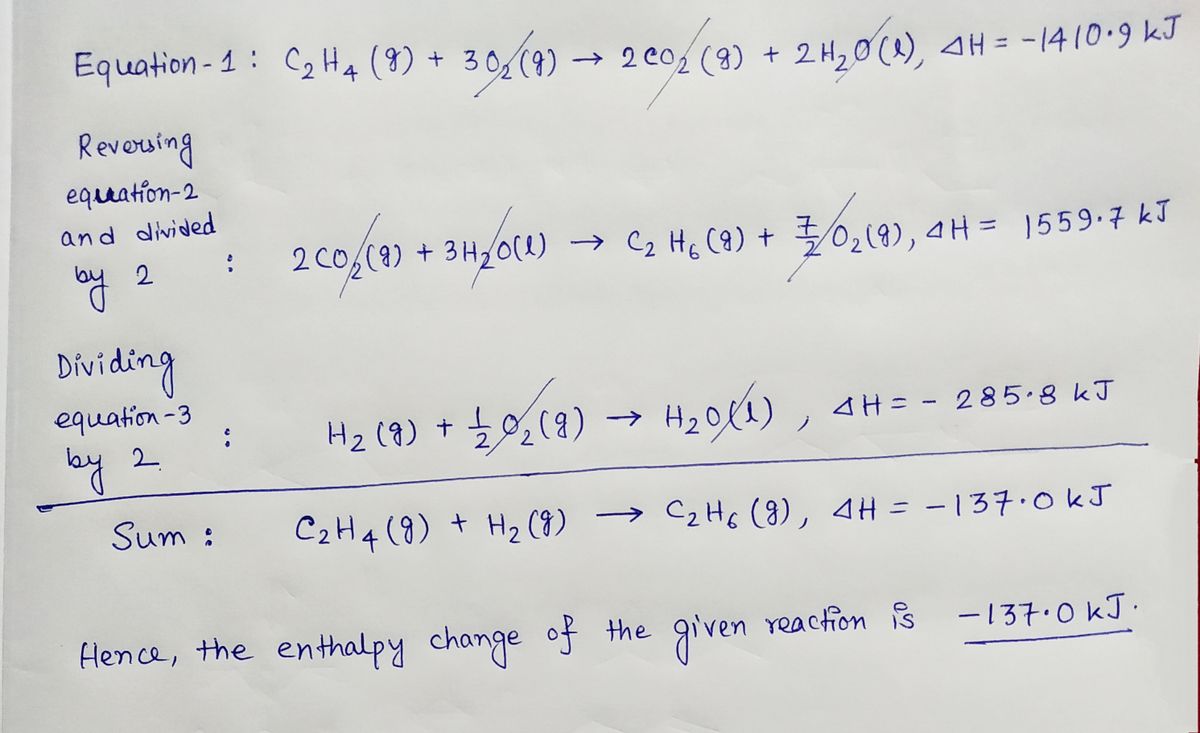
Answered Calculate the enthalpy change for the bartleby
Be sure to answer all parts. Calculate the heats of combustion for
ntif enthalpies of formation for c2h4 g co2 g and h2o l at
Calculate the value of Delta U the following reaction C 2 H 4 g
Solved Calculate the enthalpy change for the reaction Chegg
Solved 2. Calculate the enthalpy change for the following Chegg
Calculate the Enthalpy of reaction Hrxn Image src
For the reaction C2H4 3O2 2CO2 2H2O deltaE 1415kJ.The deltaH at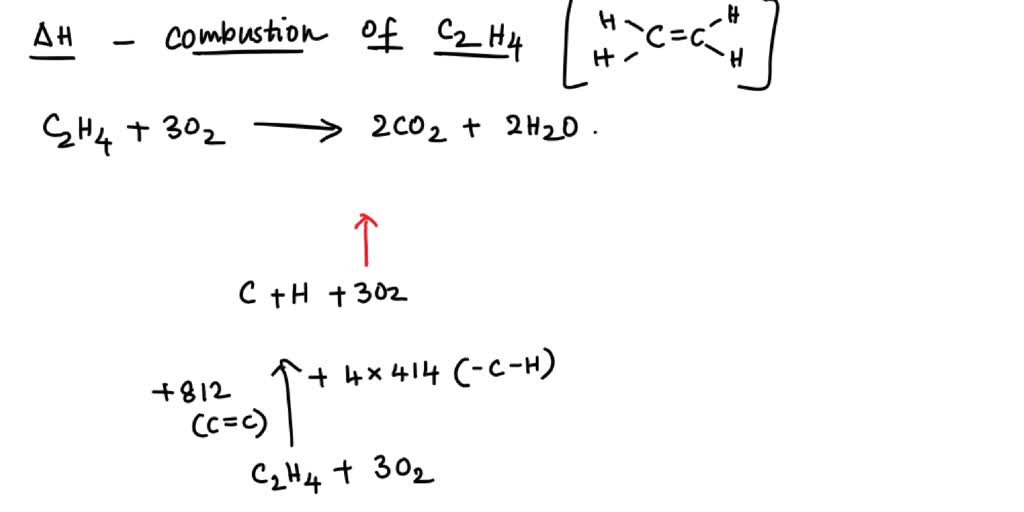
SOLVED Using the bond enthalpy values provided calculate the
THERMOCHEMISTRY Thermodynamics ppt download
Solved Hess s Law a Given the following data C2H4 g Chegg
SOLVED Calculate the heats of combustion for the following
Be sure to answer all parts. Calculate the heats of combustion for
Be sure to answer all parts. Calculate the heats of combustion for
Answered The standard enthalpy of combustion of bartleby
For complete combustion of ethene. C2H4 g 3O2 g 2CO2 g
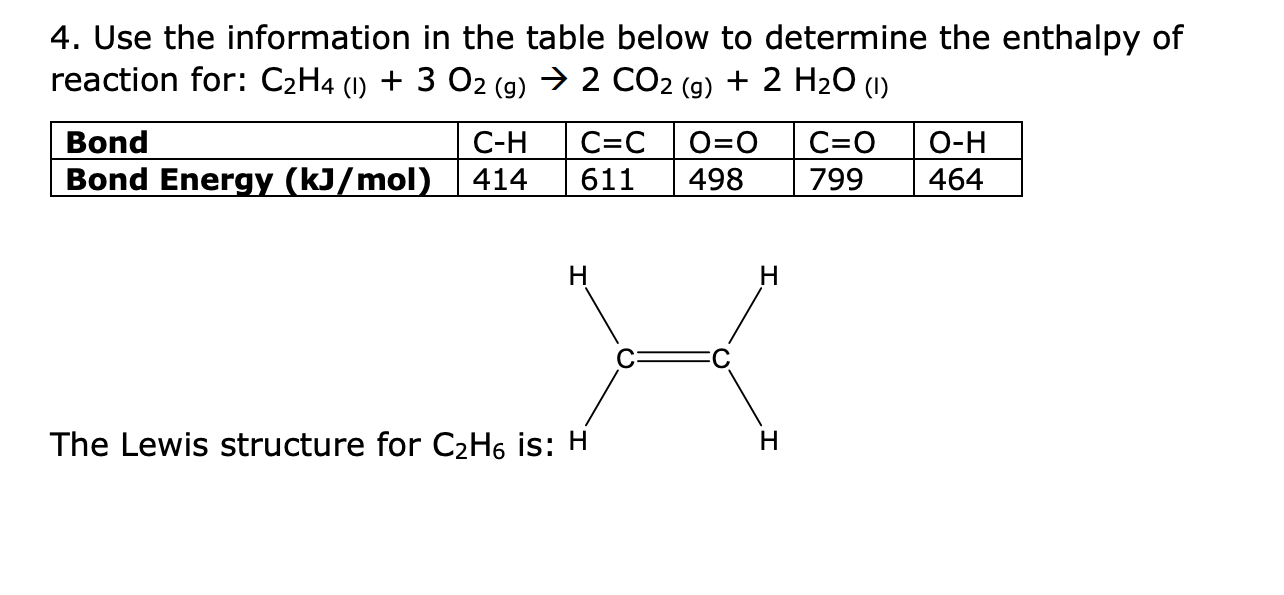
Solved 4. Use the information in the table below to Chegg
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol
Hess s Law. ppt download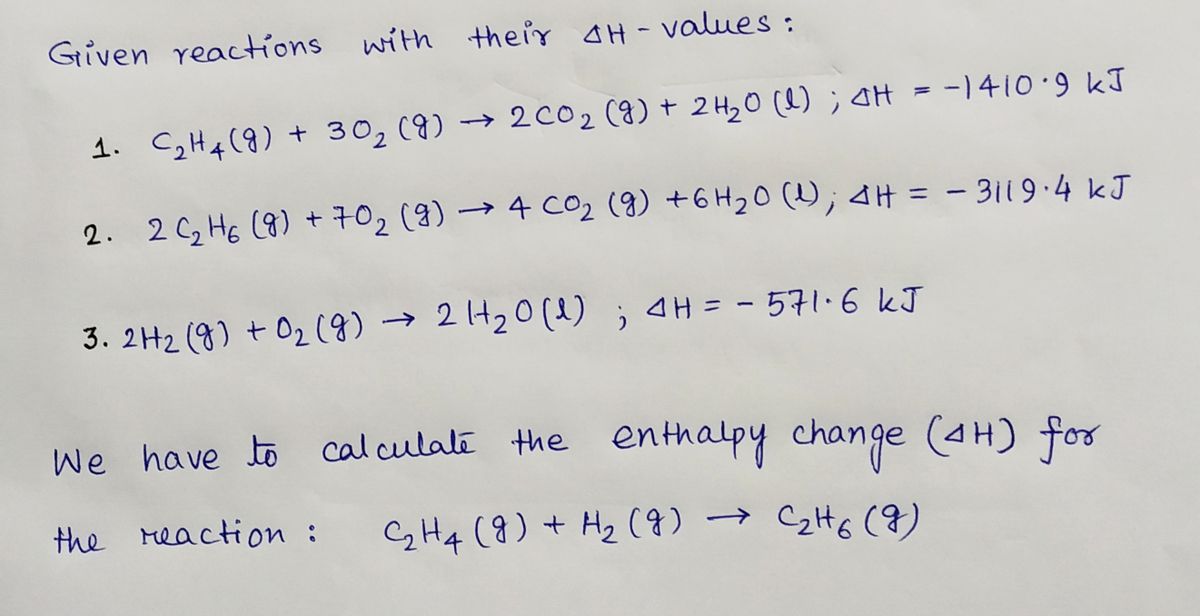
Answered Calculate the enthalpy change for the bartleby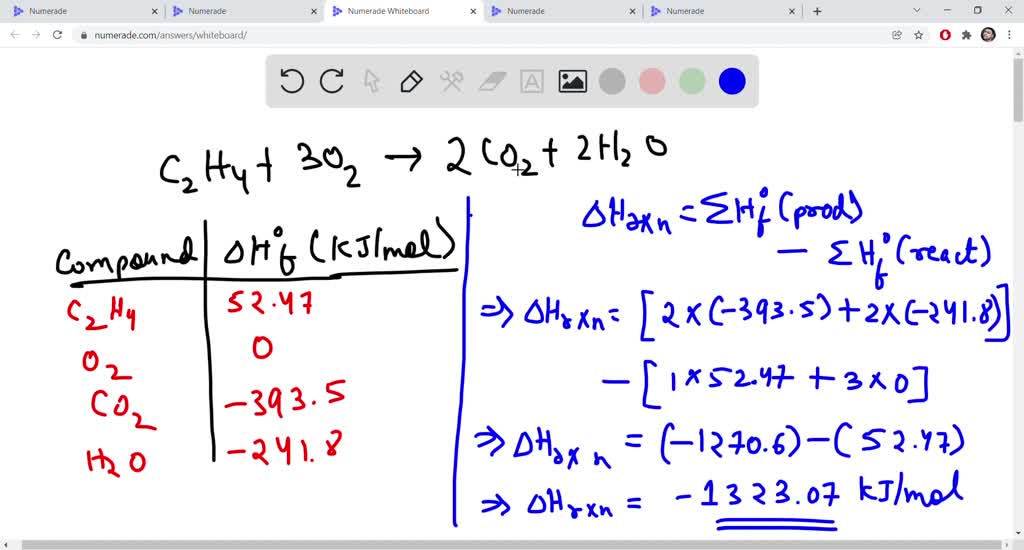
SOLVED The combustion of ethene C2H4 occurs via the reaction
Solved Given the following data C2H4 g 302 g 2 CO2 g
Solved Given the following data calculate the change in Chegg
Solved For the following Chegg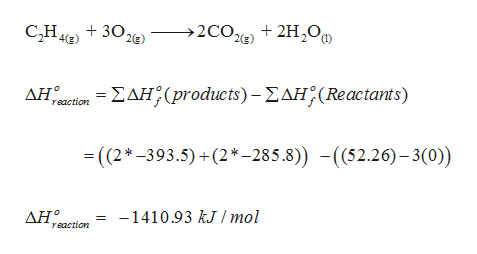
Answered Calculate the heats of combustion for bartleby
Answered The combustion of C2H4 g is bartleby
Determine the enthalpy change ofC2H4 3 O2 2 CO2 2 H2O H
Solved C2H4 g 3O2 g 2CO2 g 2H2O l Given the reaction
Answered For the combustion reaction of ethylene bartleby
7.65c Calculate the enthalpy change from bond energies C2H4 g 3O2 g 2CO2 g 2H2O g
For complete combustion of ethane C2H4 g 3O2 g 2CO2 g 2H2O l the amount of heat produce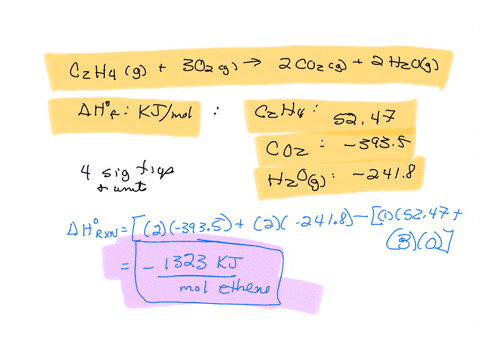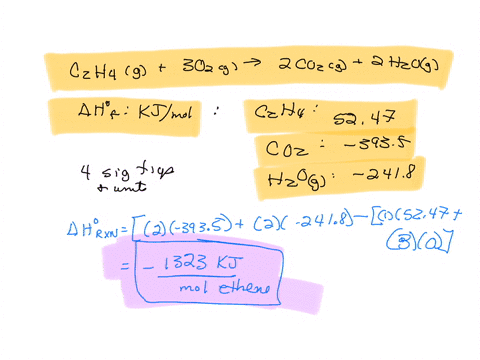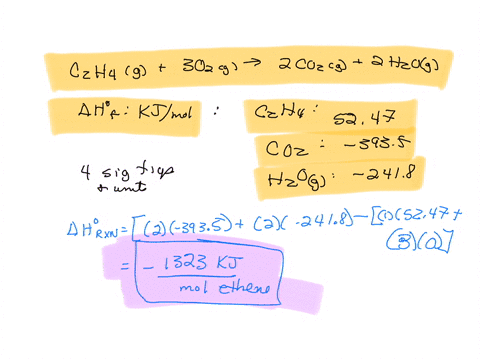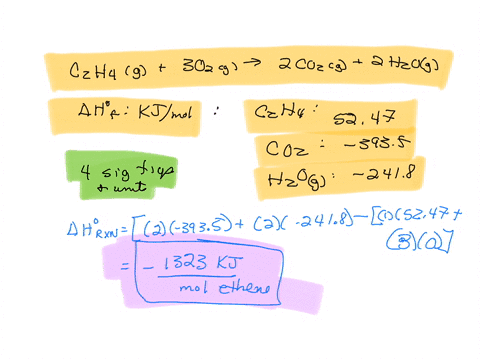
SOLVED The combustion of ethene C2H4 occurs via the reaction
Calculate the heats of combustion for the following reaction Quizlet