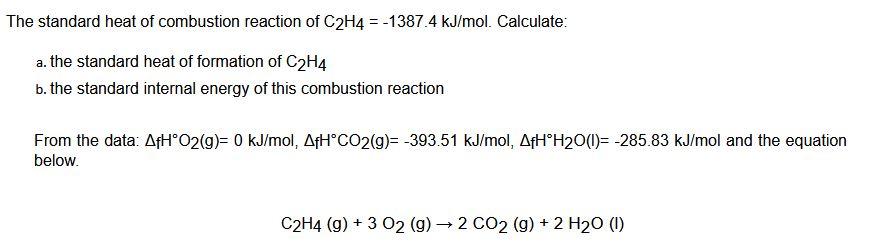Heats of combustion of C 2 H 4 H 2 and C 2 H 6 are
Heat of hot sale combustion of c2h4
Share. Visit »
Calculate the enthalpy of combustion of ethylene 1 atm pressure
The heat of combustion of C H C H and H are 1409 kJ mol 1558
Solved 1. The combustion of ethane C2H4 is an exothermic
calculate the heat of combustion of ethene Brainly.in
Solved b The enthalpies of combustion for ethene C2H4 Chegg
ntif enthalpies of formation for c2h4 g co2 g and h2o l at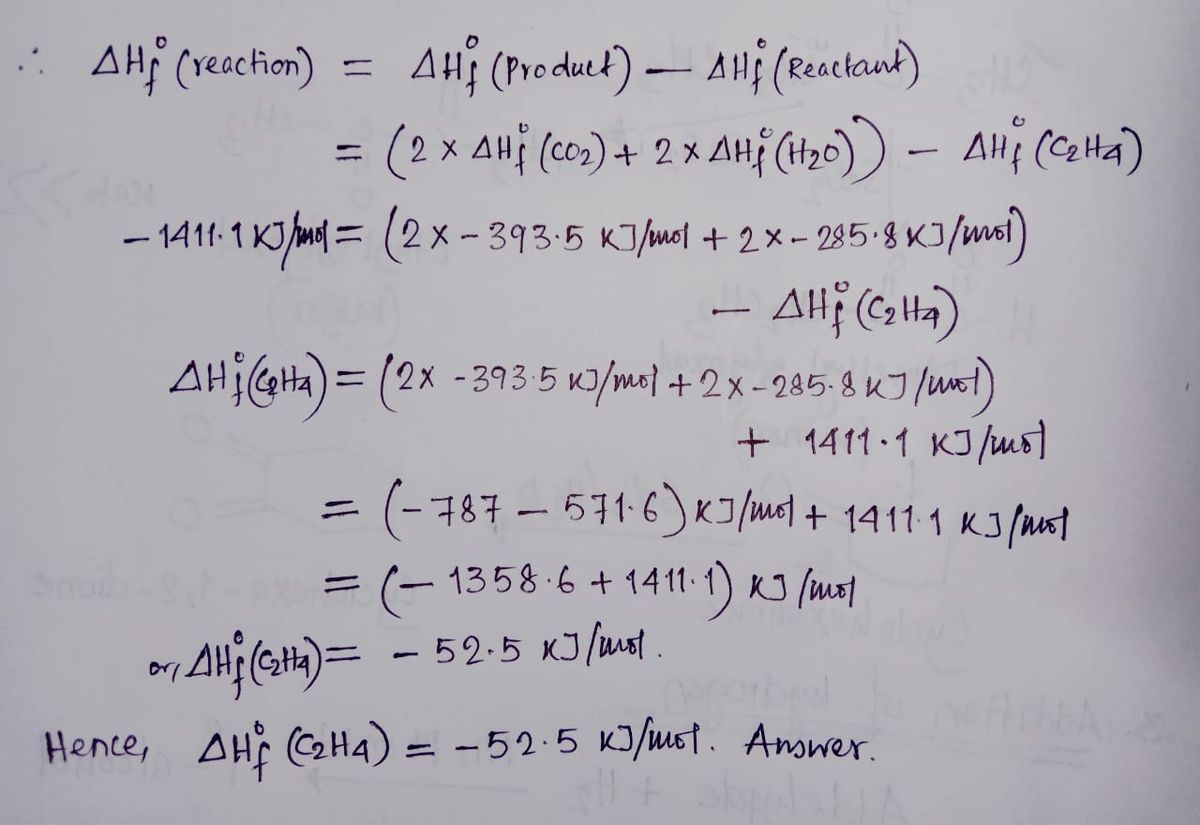
Answered The standard enthalpy of combustion of bartleby
Be sure to answer all parts. Calculate the heats of combustion for
heat of combustion of C2H4 is 337 kcal if 5.6 lit o2 is used at
Solved Using the following data determine the heat of Chegg
Answered For the combustion reaction of ethylene bartleby
Solved The standard enthalpy of combustion of ethene gas C2H4 g
Answered 5. Write a balanced thermochemical bartleby
SOLVED The standard enthalpy of combustion of ethene gas C2 H4
39 The heat of combustion of ethene C.H is 1409.3 kJ mol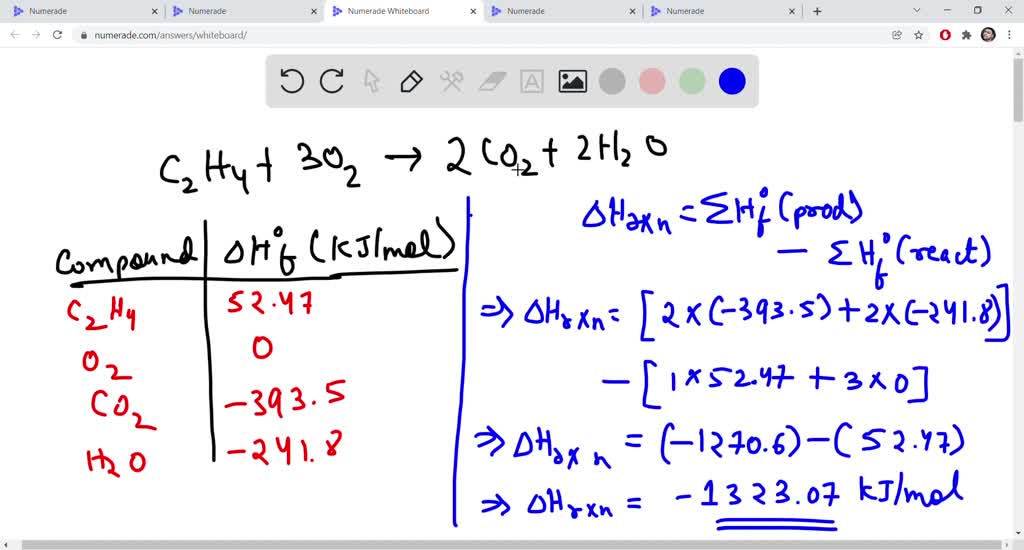
SOLVED The combustion of ethene C2H4 occurs via the reaction
heat of combustion of C2H4 is 337 k.cal if 5.6lits O2is used at
The heat of combustion of C2H6 is 368 4kcal calculate heat of
Can you explain the answer of this question below If enthalpies of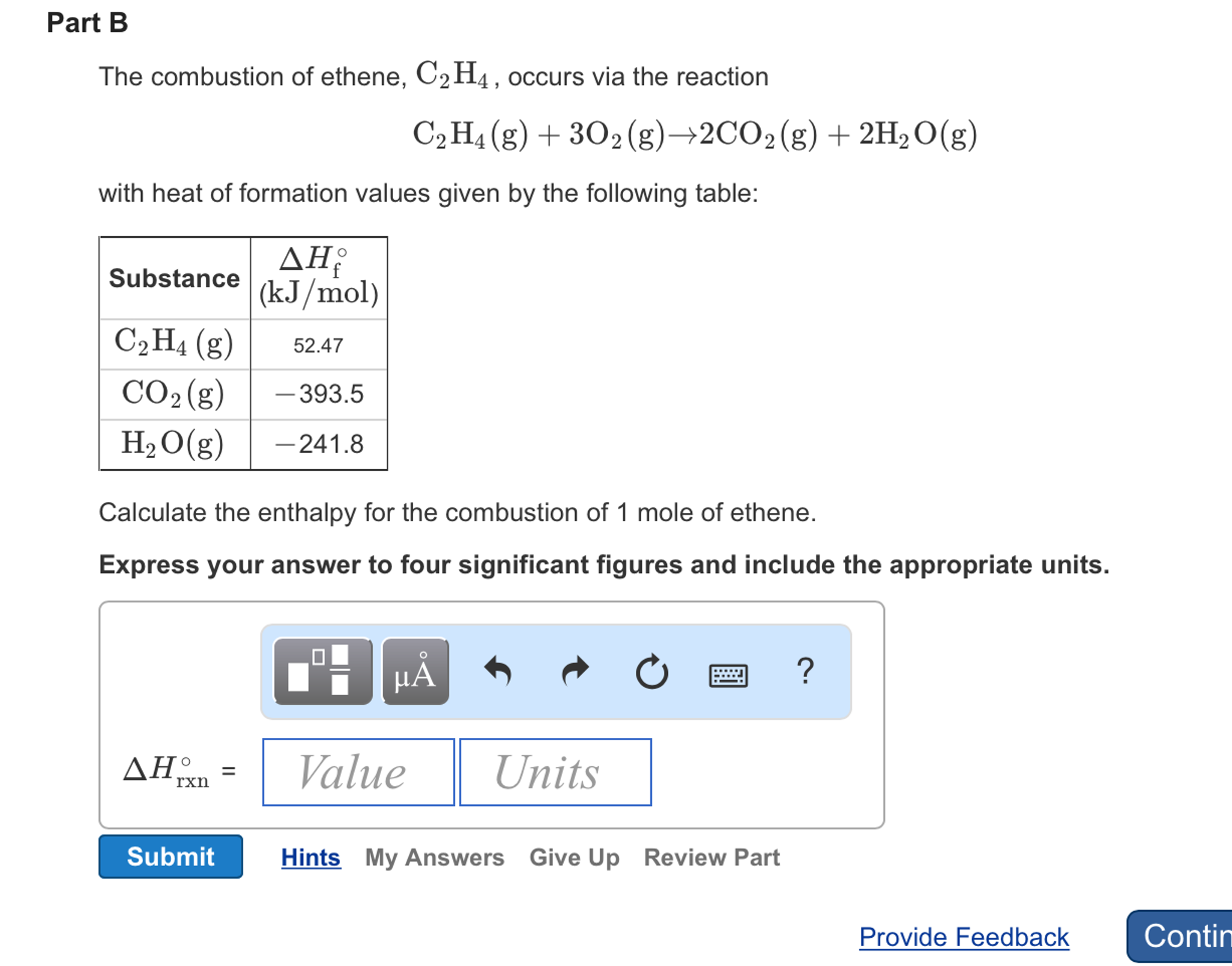
Solved The combustion of ethene C 2H 4 occurs via the Chegg
Uncal 39. The heat of combustion of ethene C H. is 1409.3 kJ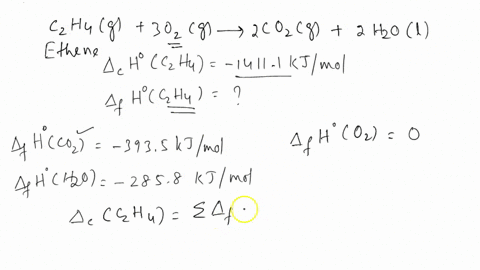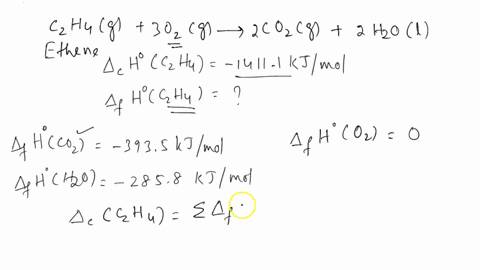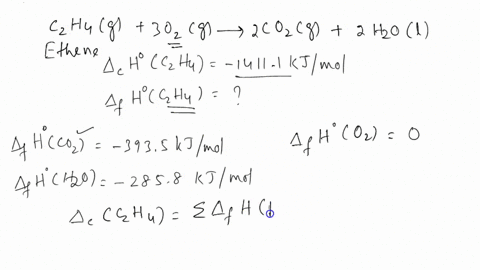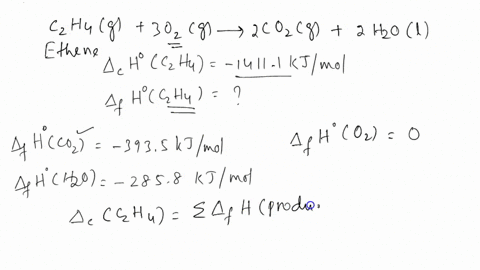
SOLVED The standard enthalpy of combustion of ethene gas C2H4 g
Use bond energies to confirm that the complete combustion of
Heat of combustion of C 2H 4 is 337 K Cal. If 5.6 lit O 2 is used
Calculate the ethalpy of combustion of ethylene g to form CO 2 gas and H 2 O gas at
In C2H4 formation of C C and C C is 590 kJ mole and 331
Solved The combustion of ethane C2H4 occurs via the Chegg
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g
The enthalpy of formation for C 2 H 4 g CO 2 g and H 2 O l at 25 C and 1 atm. Pressure be 52 394 and 286 kJ mol 1 respectively. The enthalpy of combustion of
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol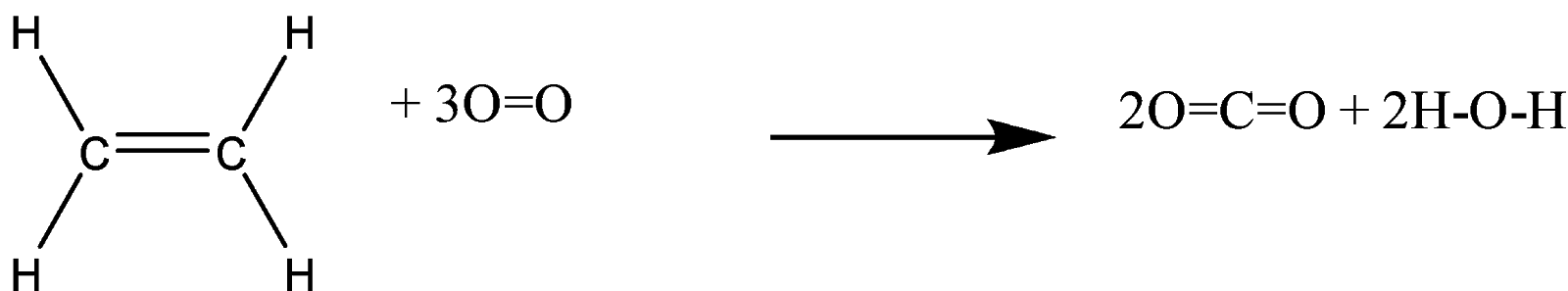
Calculate heat of combustion of ethene from bond energy data C C
31. Calculate the heat of combustion of ethene CH2 CH2 g 3O2 g
Balancing the Equation the Combustion of Ethene C2H4
Solved The standard heat of combustion reaction of C2H4 Chegg
The heat of combustion of C2H4 C2H6 and H2 are 1409.5 kJ 1558.3kJ a
Be sure to answer all parts. Calculate the heats of combustion for
Heat of combustion of CH4 C2H4 C2H6 are 890 1411 1550 kJ mol
The heats of formation of CO 2 H 2O and C 2H 4 are 94.05 68.2 and 12.6 K.Cals mole respectively. The heat of combustion of ethylene is
Chemistry Thermochemistry 6 of 37 Enthalpy Example 2 Combustion of Ethane