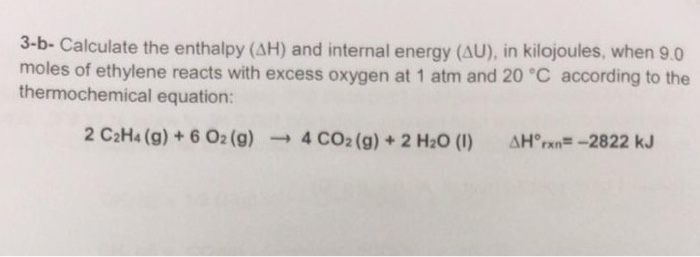
Solved 3 b Calculate the enthalpy AH and internal energy
20.0 g ethene is hot sale how many moles of c2h4
Share. Visit »
How many grams of ethylene glycol must be added to
1 mol of O2 is required to burn a mixture containing an equal
Solved 3 If 2.70 mole C2H4 reacts with 6.30 molO2 identify
SOLVED When 125.0 g of ethylene C2H4 burns in 60.0 grams of
When 100 g of ethylene polymerises entirely to polythene the weight of polyethene formed as
What is the percentage yield when 7g of ethene produces 6g of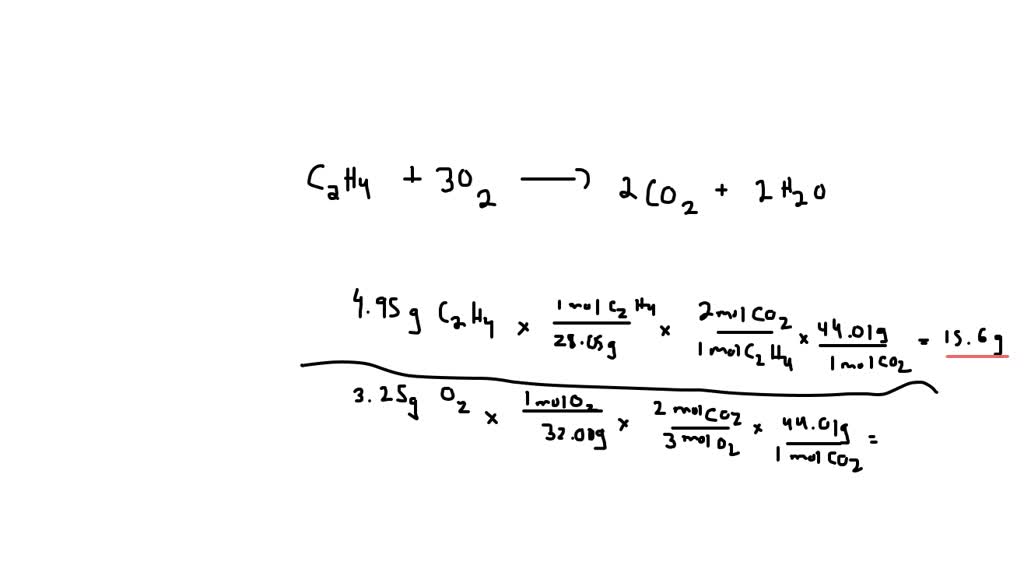
SOLVED Consider the combustion of ethylene with oxygen. C2H4 g
Consider the combustion of ethylene C2H4 3 O2 g 2 H2O g . If
What is the number of moles present in 9g of ethane Quora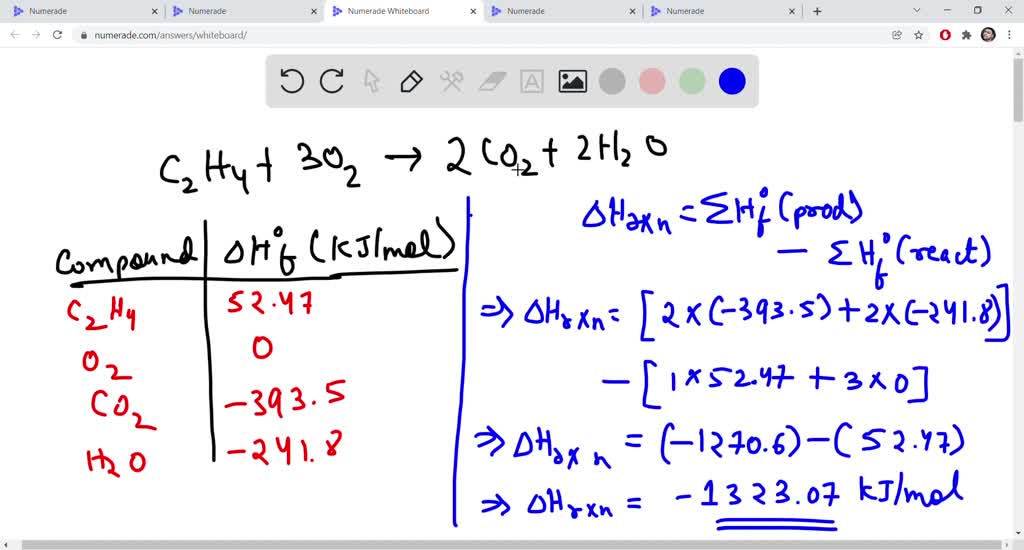
SOLVED The combustion of ethene C2H4 occurs via the reaction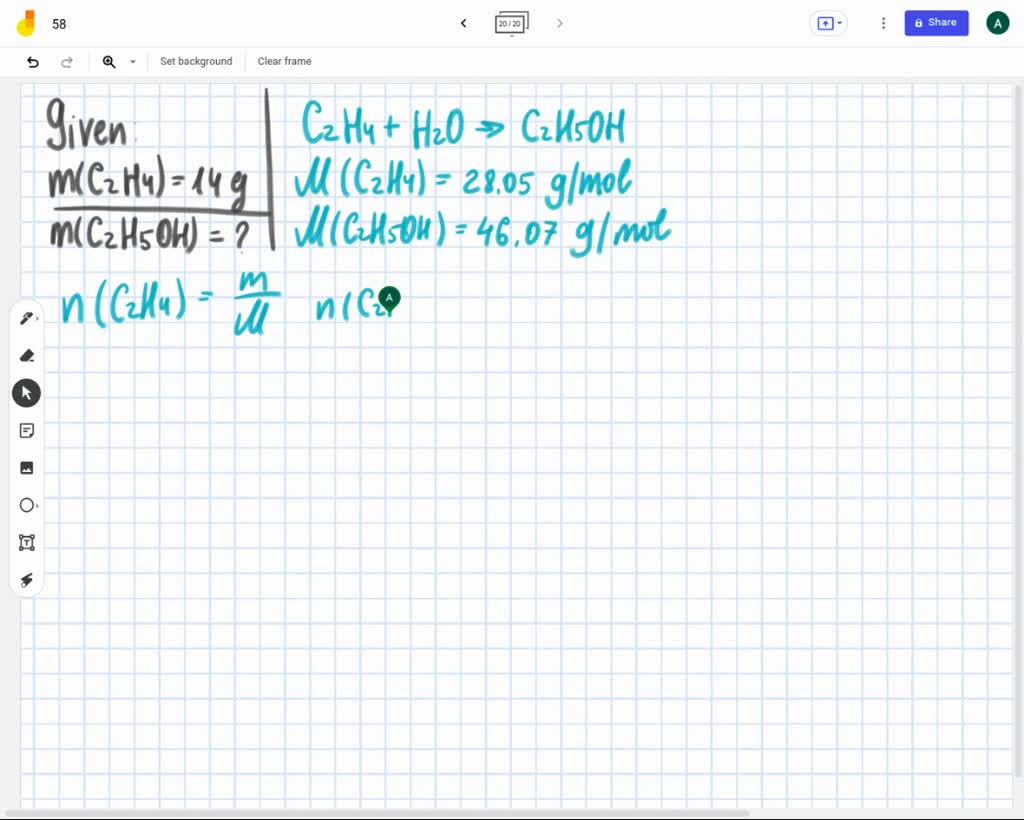
SOLVED Ethanol C 2H 6O molar mass 46.07 g mol is synthesized
Solved 28. The equation below shows the incomplete Chegg
6 The average molar mass of a mixture ofmethane CH4 and ethane
Calculate the mole fraction of ethylene glycol C 2 H 6 O 2 in a solution containing 20
Calculate the boiling point of a solution made by mixing 200 grams
Bengali What will be the number of 1 moles of ethylene 2 mole
Colligative Properties Ethylene glycol C 2H 6O 2 is the
Answered 28. The equation below shows the bartleby
Final Report PDF Cracking Chemistry Stoichiometry
How many moles of ethane are required to produce 33g of CO2 on
Which of the following is the correct set up for the problem How
C H OH C H H O 45.5 kJ mol 126 J K mol ethanol
How to Find the Number of Atoms in C2H4 Ethene
How many grams of H2O are needed to react with 3.5 moles of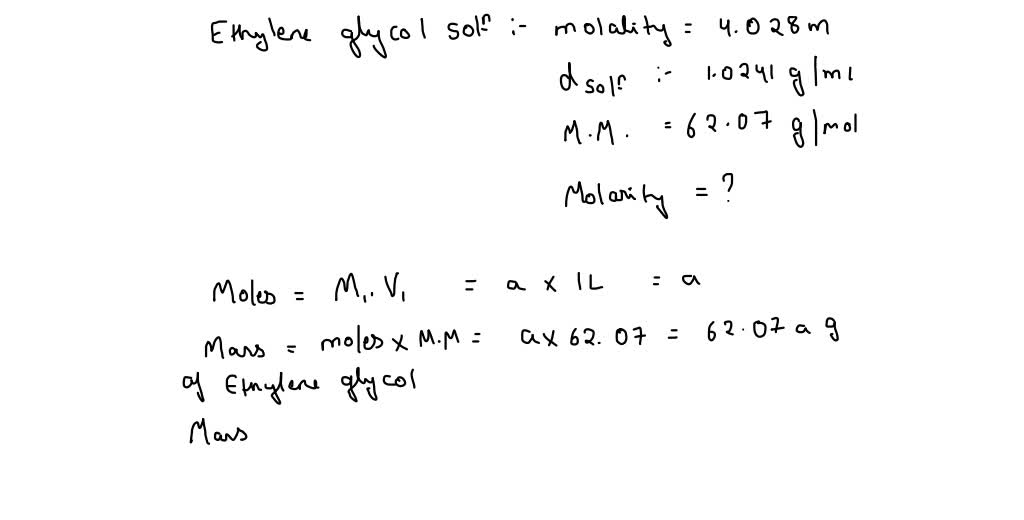
SOLVED Ethylene glycol C2H6O2 is a colorless liquid used as
Ethylene CH2 CH2 CID 6325 PubChem
Answered Ethylene oxide is prepared by oxidation bartleby
Answered 2. Ethylene burns in oxygen to form bartleby
Molar Mass Molecular Weight of C2H4 Ethene
Solved Ethene C 2H 4 can be halogenated by this reaction
OneClass The hydrogenation of ethene C 2H 4 produces ethane
Ethene C2H4 can be halogenated by this reaction C2H4 g X2 g
Answered 28. The equation below shows the bartleby
Gas Stoichiometry Chemistry Socratic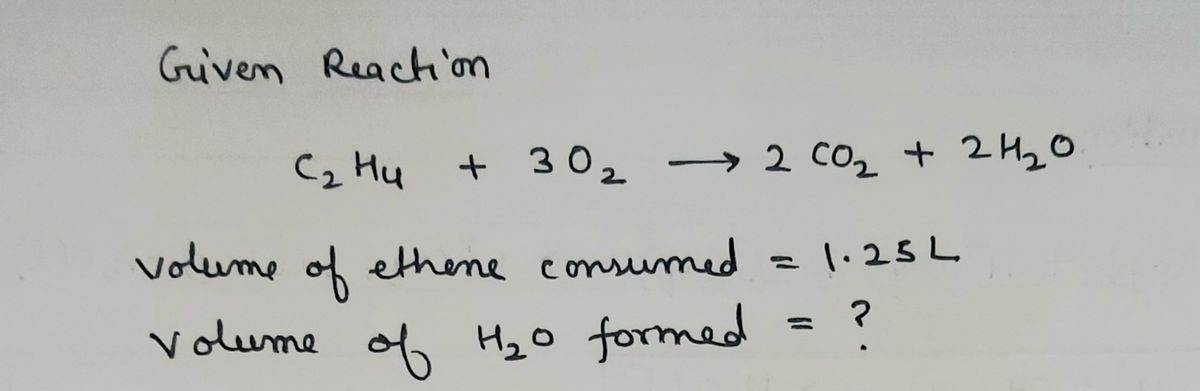
Answered 2. Ethylene burns in oxygen to form bartleby
Answered If the complete combustion of an bartleby
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol
Determine the required concentration in percent by mass for an
Which of the following compounds 1 mole of it requires 96g of



