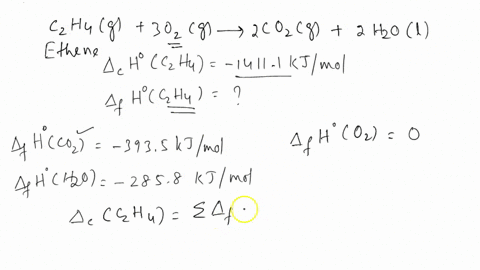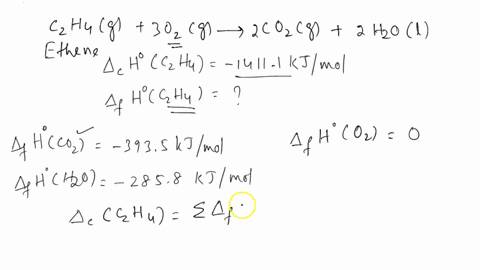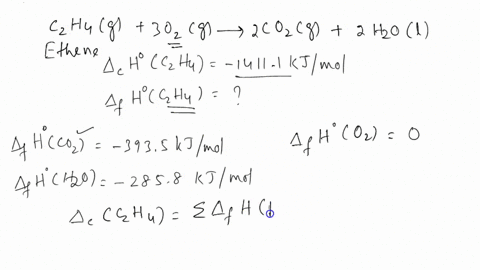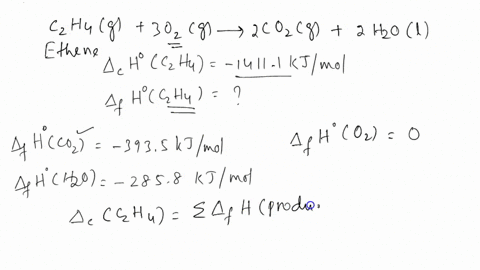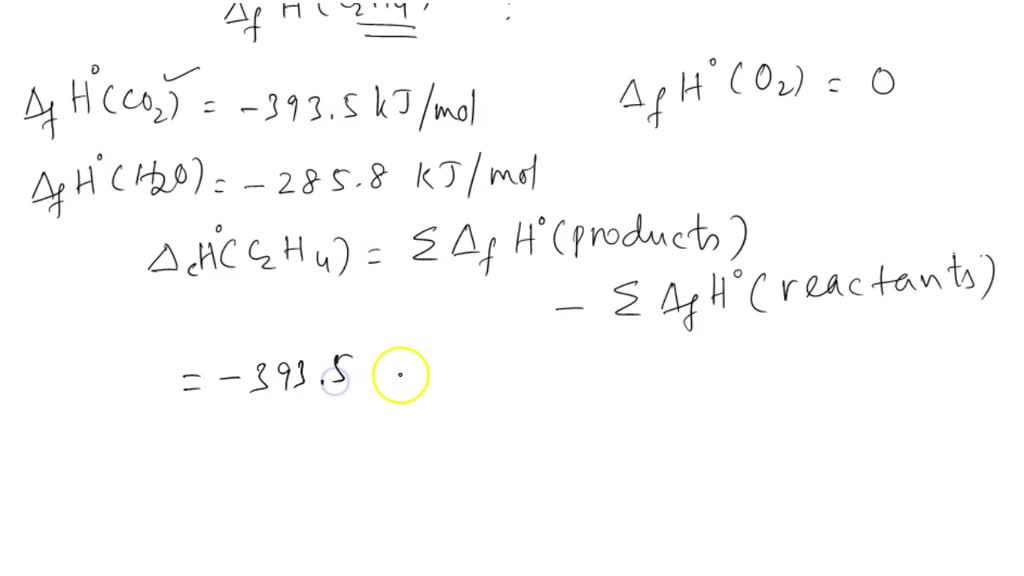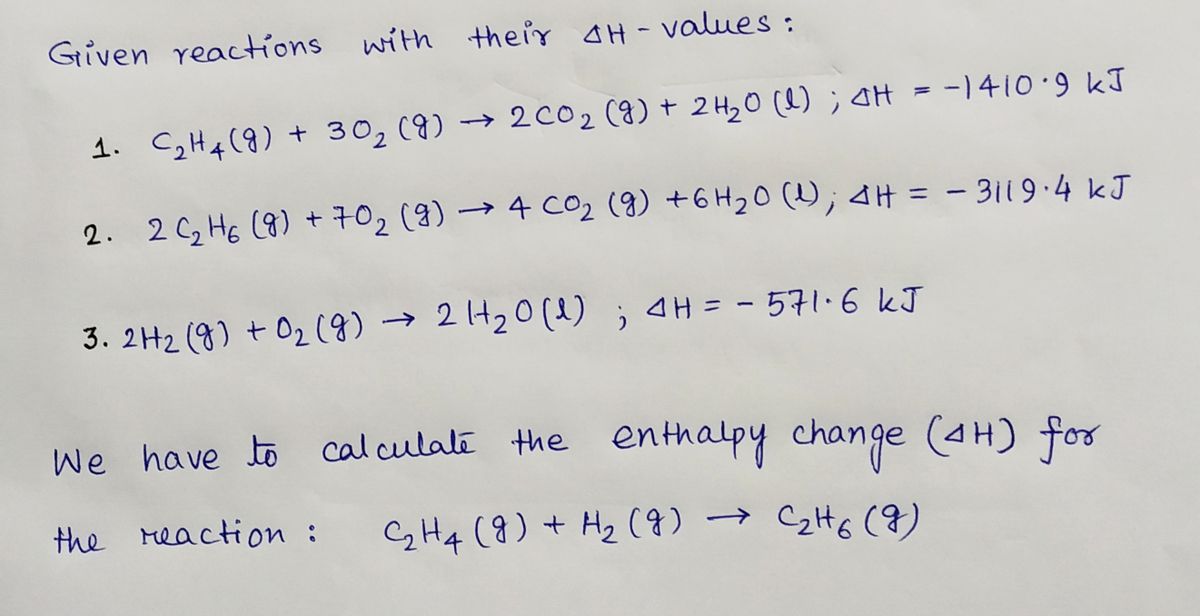ASSIGNED READINGS
Delta h hot sale formation of c2h4
Share.
Visit »

Be sure to answer all parts. Calculate the heats of combustion for 
18. Calculate the enthalpy of hydrogenation of C2H2 g to C2H4 g 
ntif enthalpies of formation for c2h4 g co2 g and h2o l at 
OneClass The combustion of ethane C2H4 occurs via the reaction 
Can you explain the answer of this question below If enthalpies of 
Be sure to answer all parts. Calculate the heats of combustion for 
Q If enthalpies of formation for C2H4 g CO2 g and H2O l at 25 
Solved Sample Question 8 What is the standard enthalpy of Chegg 
calculate the standard enthalpy of formation of ethene C2H4 from 
Calculate the ethalpy of combustion of ethylene g to form CO 2 gas and H 2 O gas at 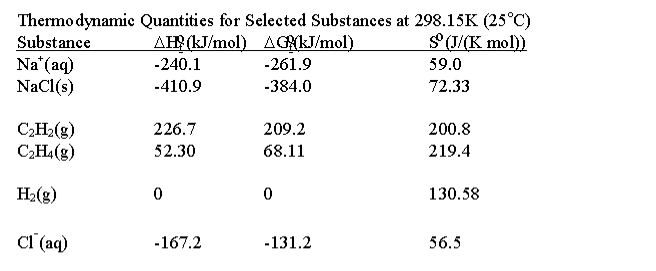
Another Thermo Question Socratic 
39 The heat of combustion of ethene C.H is 1409.3 kJ mol 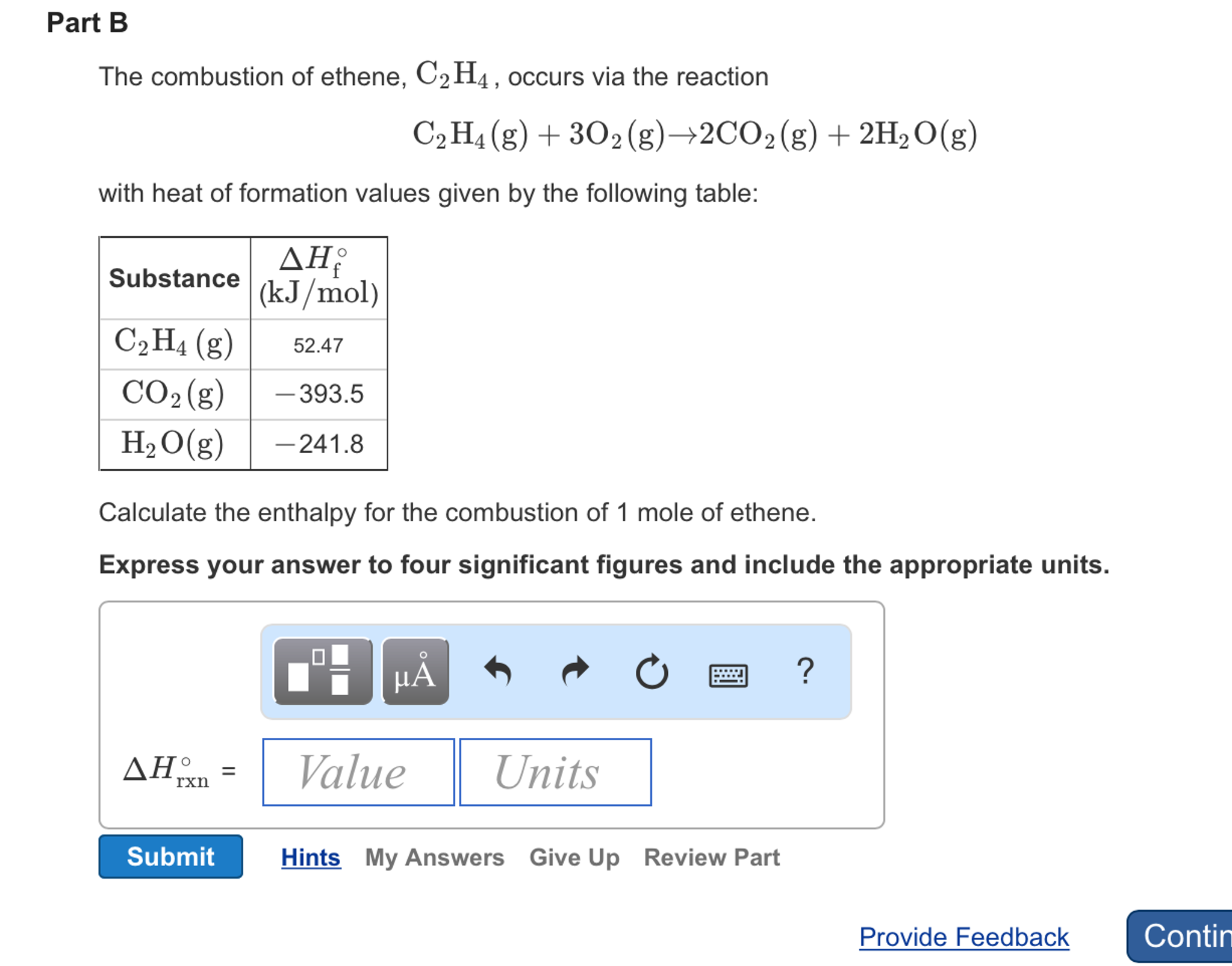
Solved The combustion of ethane C 2H 4 occurs via the Chegg 
3.Calculate the standard enthalpy of formation of C2H4 g from the 
Calculate enthalpy formation of ethylene from the following data 
Solved 17. Find the standard enthalpy of formation of Chegg 
Calculate the enthalpy of combustion of ethylene 1 atm pressure 
If f H C2H4 f H C2H6 are X1 X2 Kcal mol then heat of 
SOLVED The standard enthalpy of combustion of ethene gas C2 H4 
Solved What is the standard heat of formation for ethylene Chegg 
Calculate the enthalpy of formation of ethane from the following data i C s O 2 g to C 
If enthalpies of formation for C2H4 g CO2 g and H2O l at 25 C 
Hf C2H4 12.5 kCal Heat of atomisation of C 171 kCal Bond 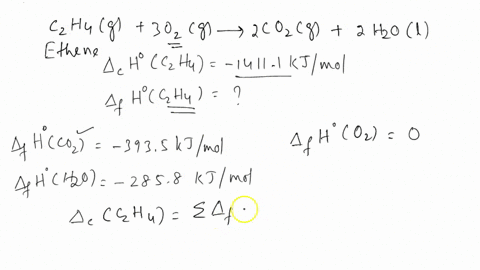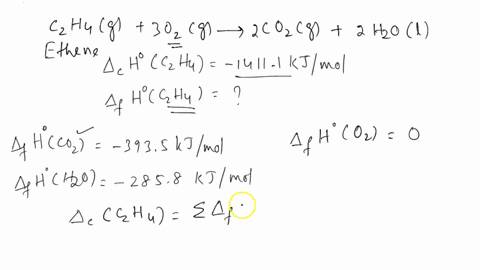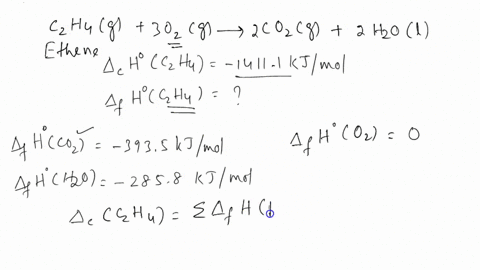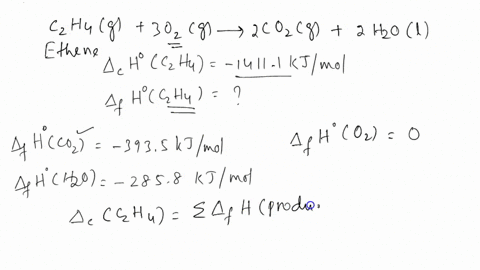
SOLVED The standard enthalpy of combustion of ethene gas C2H4 g 
If Delta f H circ C 2 H 4 and Delta f 
Solved Calculate the standard enthalpy of formation of Chegg 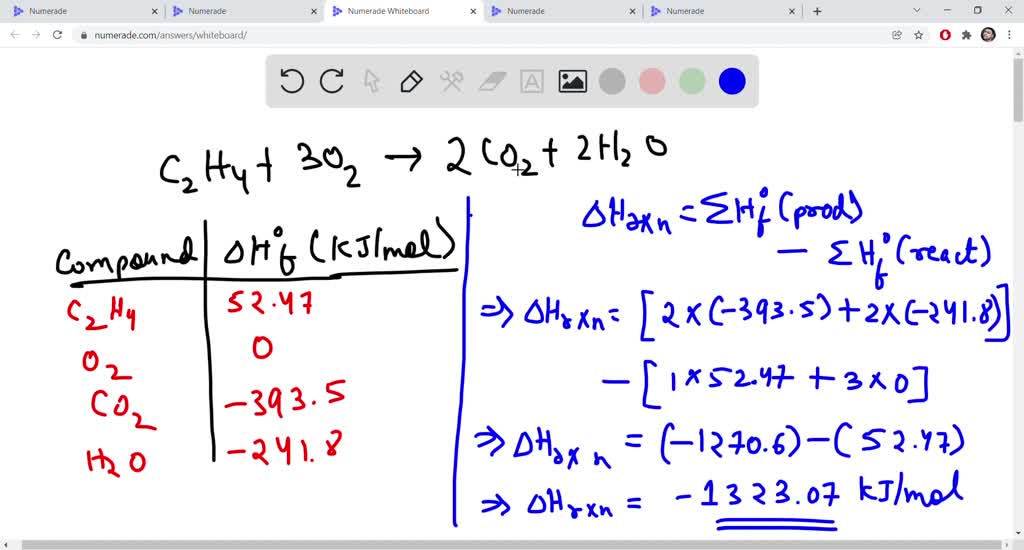
SOLVED The combustion of ethene C2H4 occurs via the reaction 
Hf C2H4 12 5 kcal Heat of atomisation of C 171 kcal Bond 
Solved Question 10 Find the standard enthalpy of formation Chegg 
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g 
Be sure to answer all parts. Calculate the heats of combustion for 
24. Delta Hf C2H4 12.5 kcal Heat of atomization of c 171 kcal 
Solved 9. Calculate the heat of formation for ethane C2H4 g 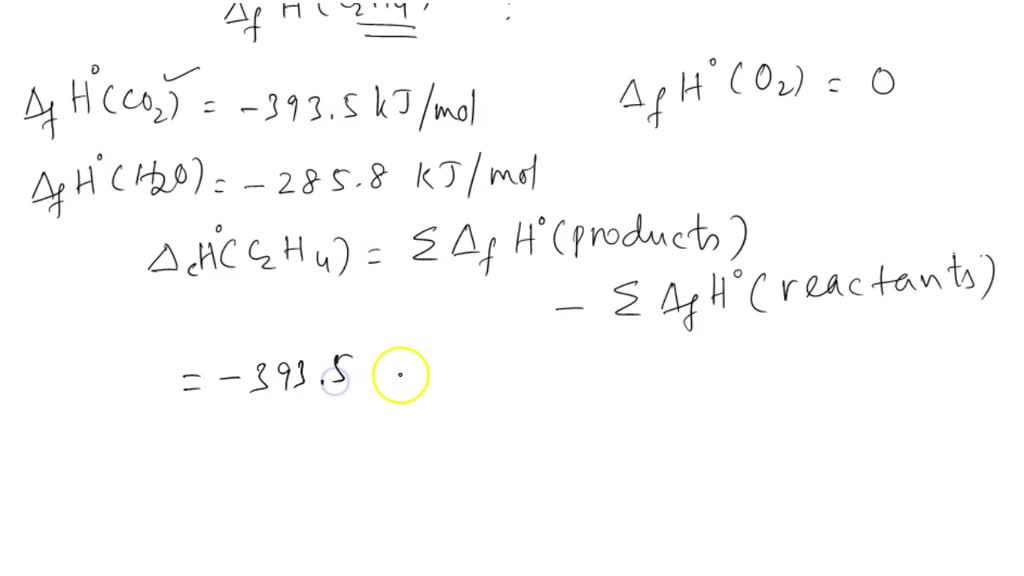
SOLVED The standard enthalpy of combustion of ethene gas C2H4 g 
Answered What is the enthalpy change of the bartleby 
In C2H4 formation of C C and C C is 590 kJ mole and 331 
A 95.4 kJ B 95.4 kJ C 141.5 kJ D 233.7 kJ 57. If AH 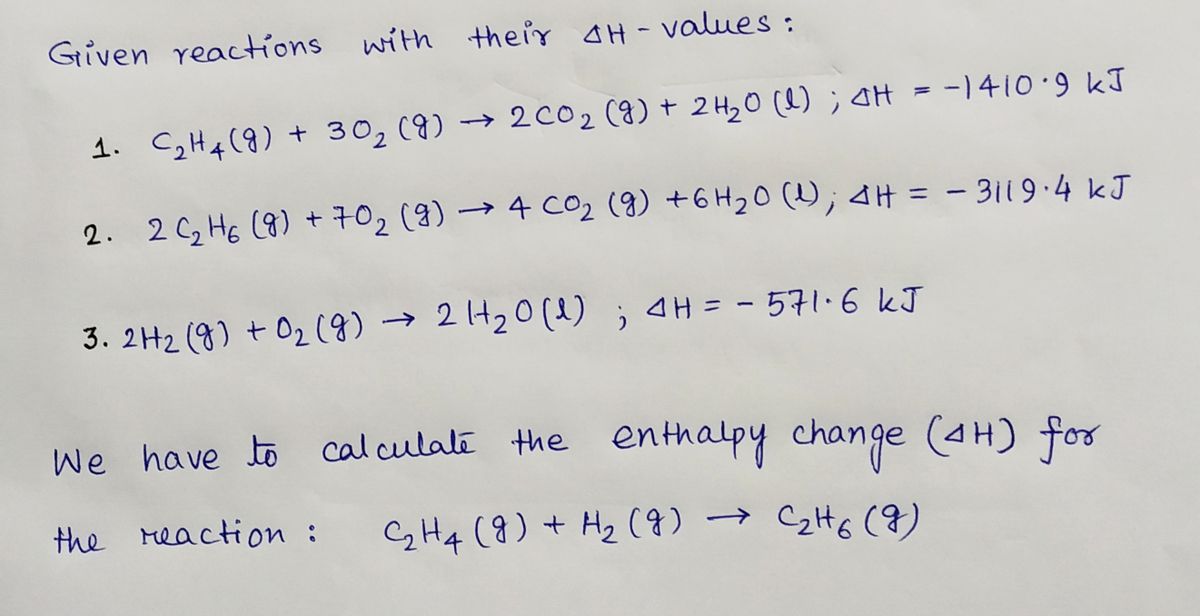
Answered Calculate the enthalpy change for the bartleby 
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol