
Answered Carbon C and oxygen gas 0 combine bartleby
C2h4 3o2 2co2 2h2o hot sale max ass of carbon dioxide
Share. Visit »
CHEMISTRY 10 UNIT 1.pptx
How to calculate the amount of carbon dioxide produced by the
Solved Ethylene oxide is produced by the catalytic oxidation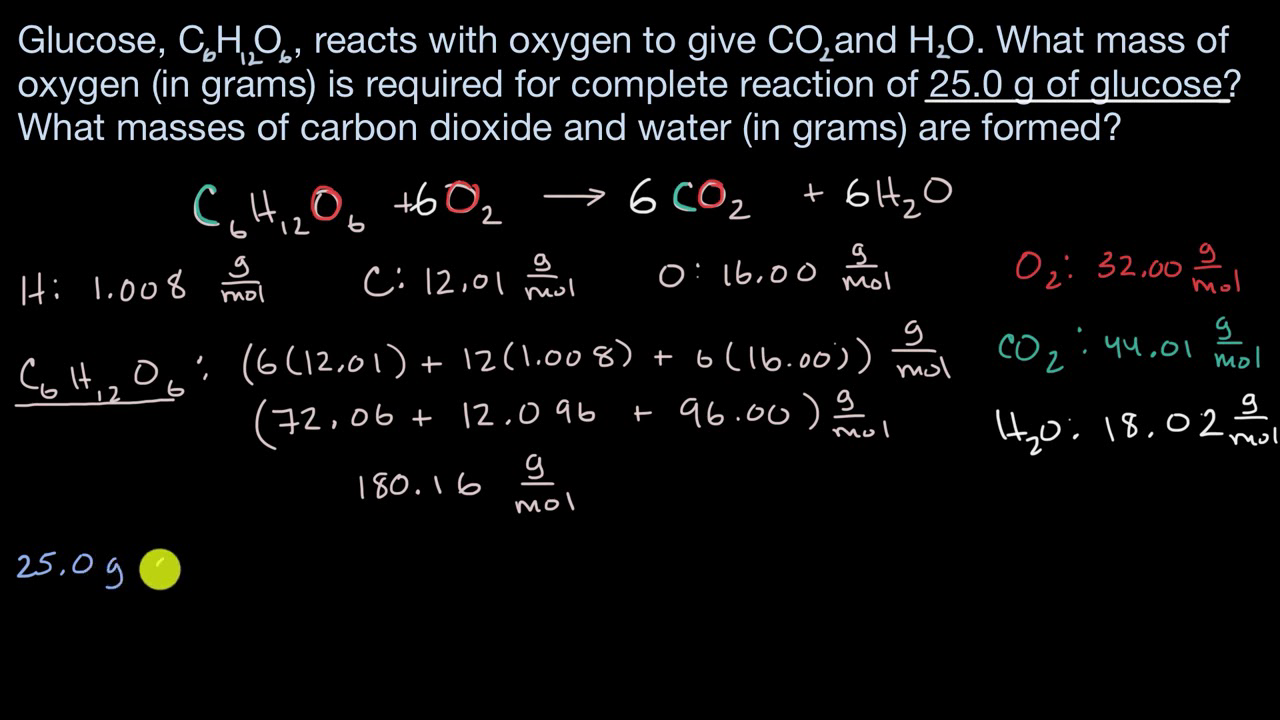
Worked example Calculating amounts of reactants and products
Answered Fermentation is a chemical process that bartleby
Carbon Monoxide in Main Group Chemistry Journal of the American
Answered CH4 2O2 2H2O CO2 what is the bartleby
Modern inorganic chemistry
The oxidation of glucose 1C6H12O62 in body tissue produces CO2 an
What mass of 2CO is produced from 14g of carbon monoxide in the
Use bond energies to confirm that the complete combustion of
An Economy Based on Carbon Dioxide and Water Potential of Large
Given the following reaction C3H8 O2 CO2 H2O what is the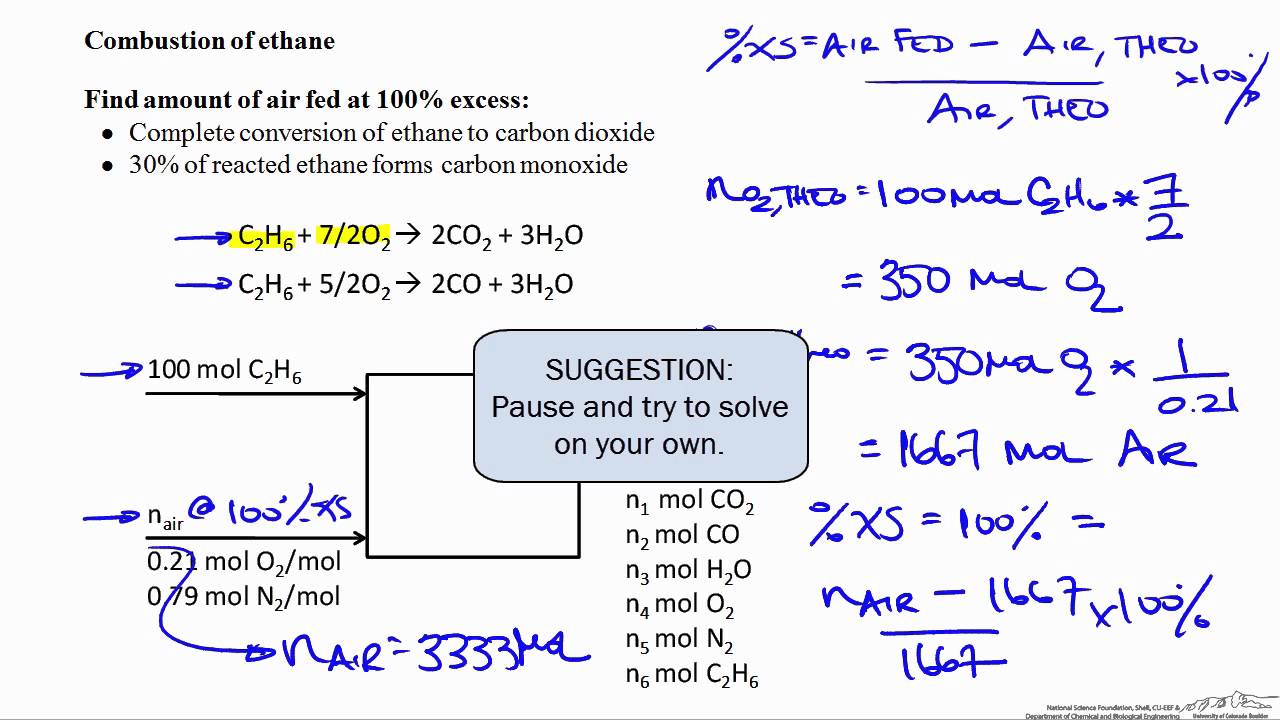
Percent Excess Air Combustion
What happens when you combine H O and CO Quora
When one mole of CO2 C C2H6 and CH4 are completely burnt in
What happens when you combine H O and CO Quora
After The Test For Extra Credit Write 10 Compound Formulas Using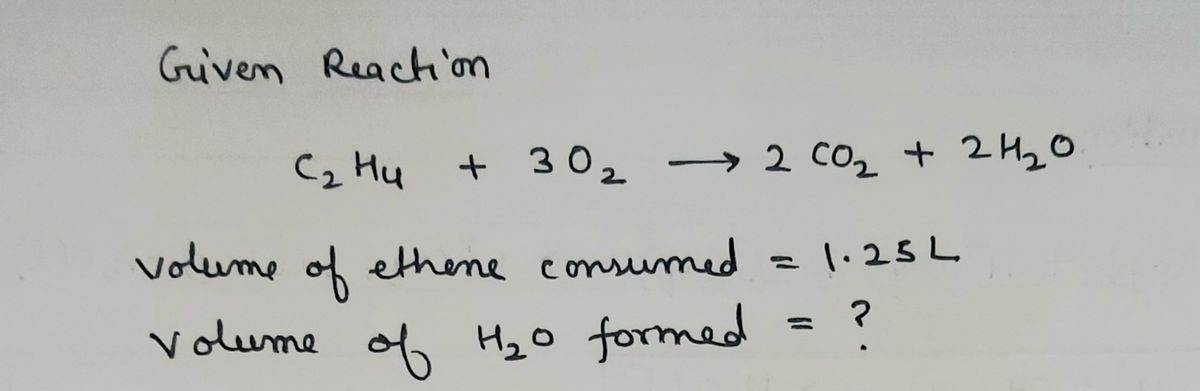
Answered 2. Ethylene burns in oxygen to form bartleby
List of chemical reactions considered for the development of, how many grams of carbon dioxide (CO2) will be produced in a combustion reaction.jpg)
The Stoichiometry of Mr.Q s Most Amazing Soup ppt download
200 ml of ethylene is burnt in just sufficient air containing 20
Mole Abc PDF Mole Unit Gases
Rebreather Wikipedia
Answered 1. The combustion of carbon monoxide bartleby
Burning is a chemical change ppt download
Carbon monoxide gas reacts with hydrogen gas to form methanol. CO
What mass of natural gas CH4 must burn to emit 267 kJ of heat
Carbon dioxide Wikipedia
Chemical kinetics PPT, how many grams of carbon dioxide (CO2) will be produced.jpg)
The Stoichiometry of Mr.Q s Most Amazing Soup ppt download
Which is the limiting reactant in the reaction of 78.0g of CH4
When burning 1200 grams of carbon how much carbon monoxide in
The combustion of gasoline produces carbon dioxide and water. Ass
Draw molecular representations of the materials before and after
Chemistry 10 12 PDF PDF Atoms Reaction Rate
Mole Ratio for CH4 O2 CO2 H2O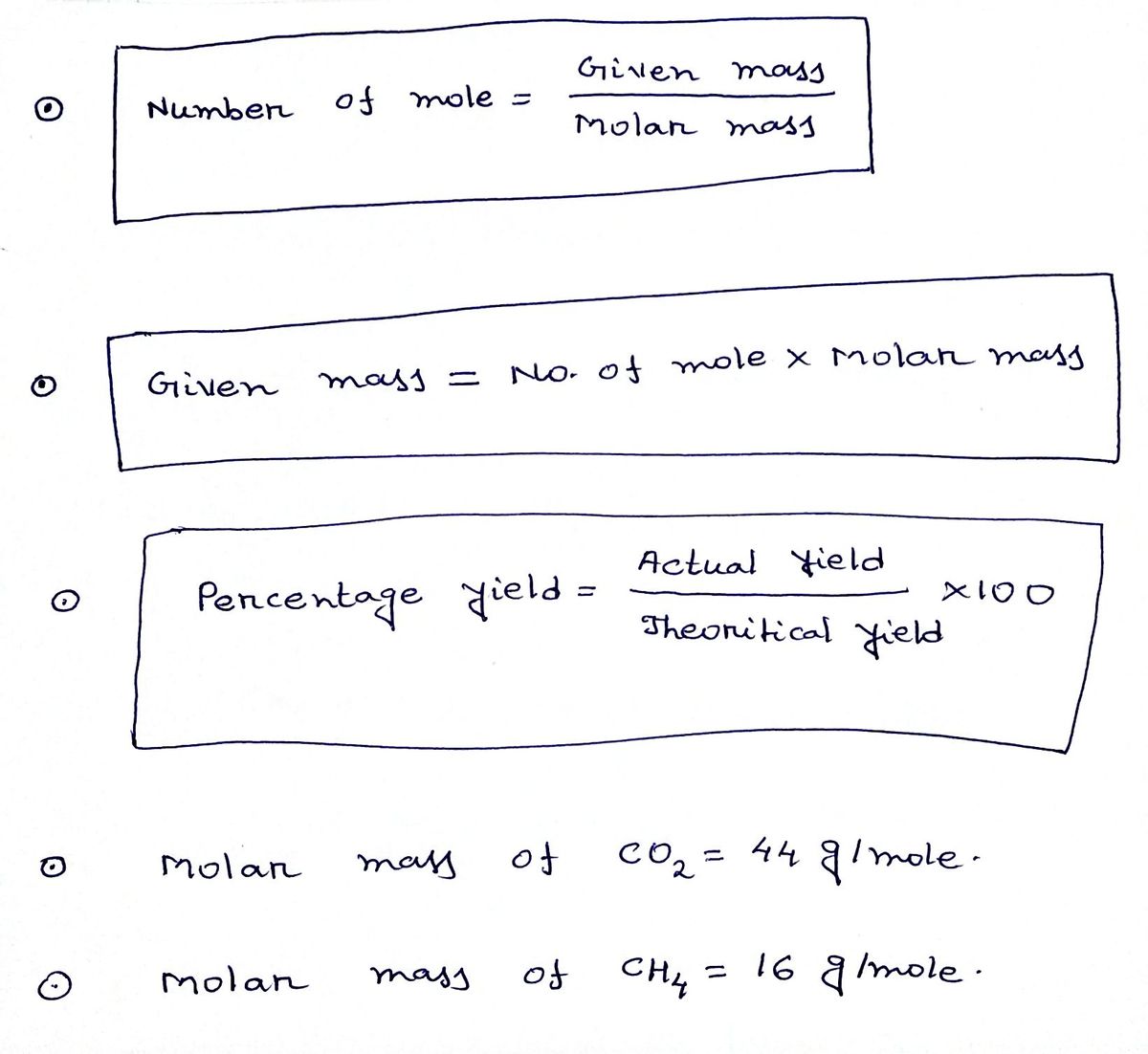
Answered CH4 2O2 2H2O CO2 what is the bartleby
What is the mass in grams of carbon dioxide that would be required




