
Be sure to answer all parts. Calculate the heats of combustion for
Delta hot sale h c2h4
Share. Visit »
3.Calculate the standard enthalpy of formation of C2H4 g from the
OneClass Now consider the following set of reactions C2H5OH
H 1C 2H 4 12.5 kcal Heat of atomisation of C 171 kcalBond energy of H 2 104.3 kcal Bond energy
Hf C2H4 12.5 kCal Heat of atomisation of C 171 kCal Bond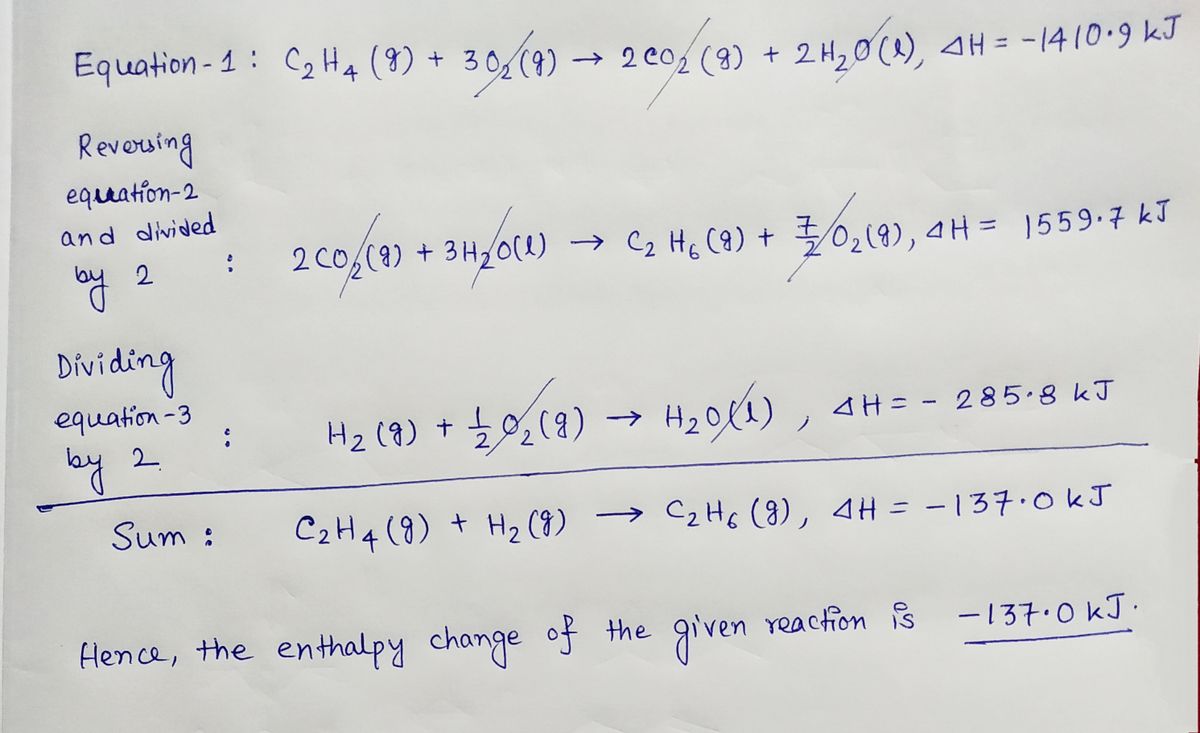
Answered Calculate the enthalpy change for the bartleby
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol
If enthalpies of formation for C2H4 g CO2 g and H2O l at 25 C
ASSIGNED READINGS
IN THE SOLUTION WHY IS THE UNDERLINED STEP USED H reactants H
Heats of combustion of C 2 H 4 H 2 and C 2 H 6 are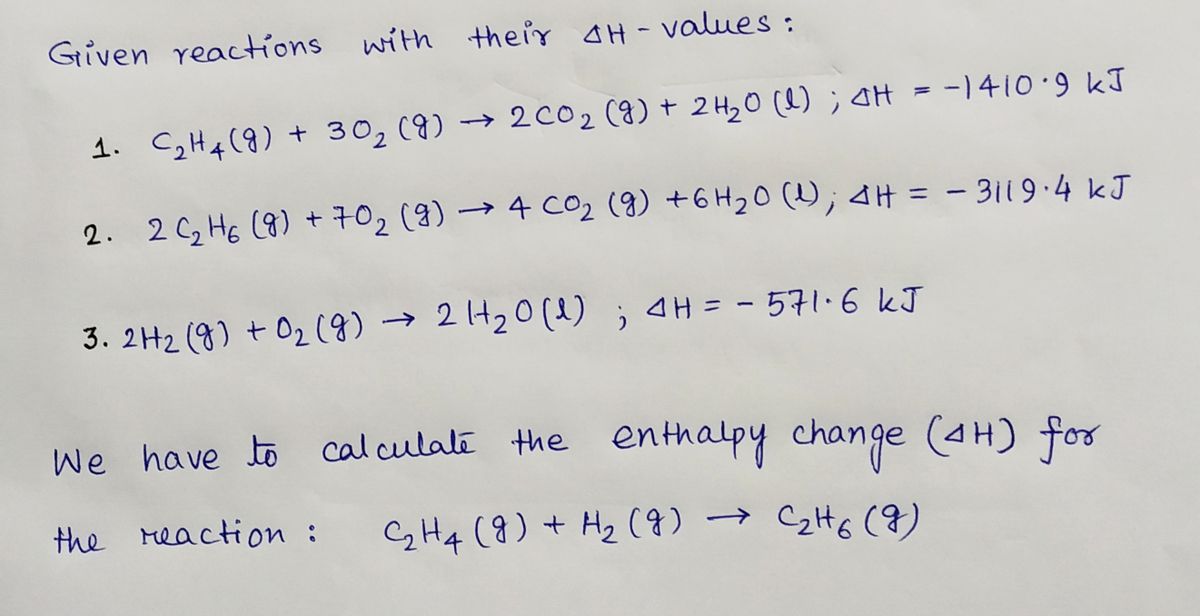
Answered Calculate the enthalpy change for the bartleby
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g
Hf C2H4 12 5 kcal Heat of atomisation of C 171 kcal Bond
Solved Calculate the enthalpy change for the reaction Chegg
Diketahui delta Hd C2H4 g 52 5 kj mola. Tuliskan persamaan
Solved Question 1 Calculate delta H for the reaction Chegg
Delta H 1 C 2 H 4 12.5 kcal Heat of atomisation of C 171 Kcal Bond energy of H 2 104
Solved Given the standard enthalpy changes for the following
Solved 4. Given delta H 52.4 kJ mol for C2H4 delta Hf Chegg
Solved calculate delta H for the reaction C2H4 g H2 g Chegg
If f H C2H4 f H C2H6 are X1 X2 Kcal mol then heat of
Use bond energies to confirm that the complete combustion of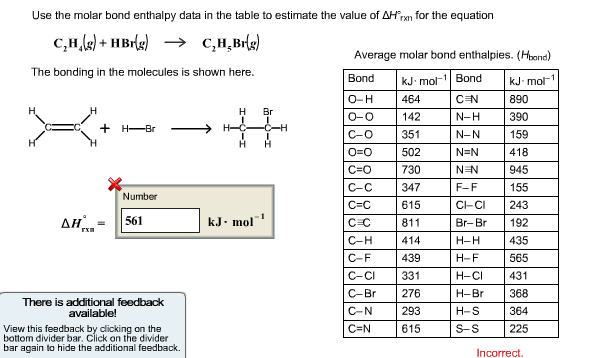
Solved use the molar bond enthalpy data in the table to Chegg
If Delta f H circ C 2 H 4 and Delta f
Solved Use bond energy values to estimate Hfor the following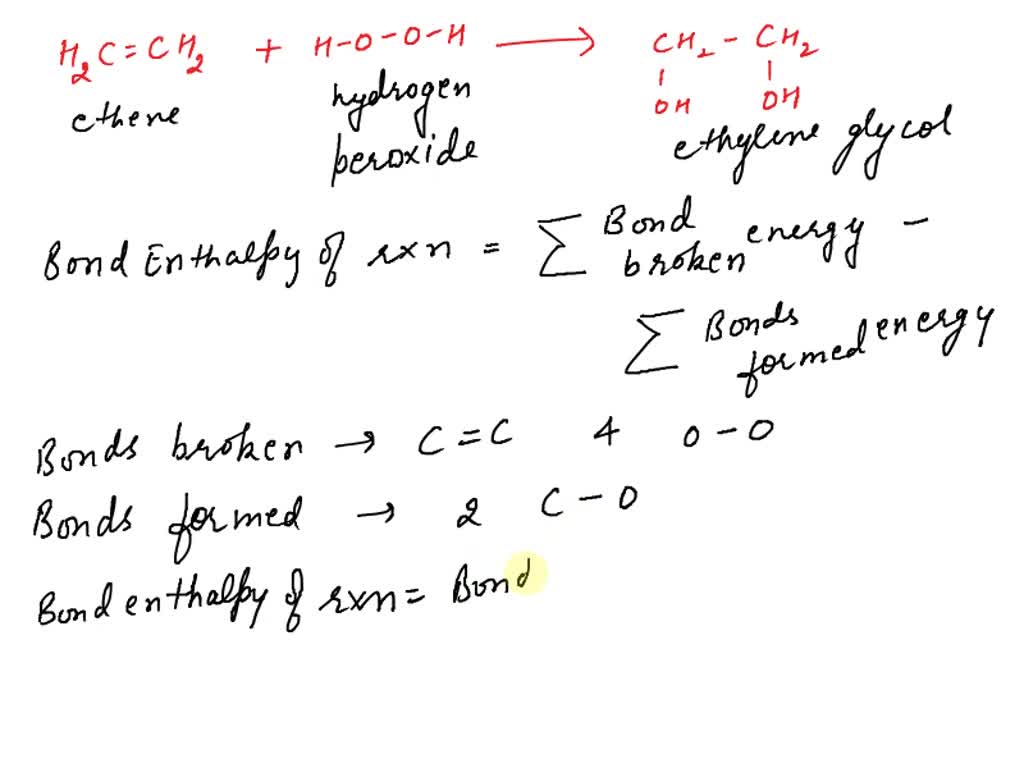
SOLVED Calculate bond enthalpy for this reaction C2H4 H2O2
Answered What is the enthalpy change of the bartleby
Chemistry 4.4 Hess s law Flashcards Quizlet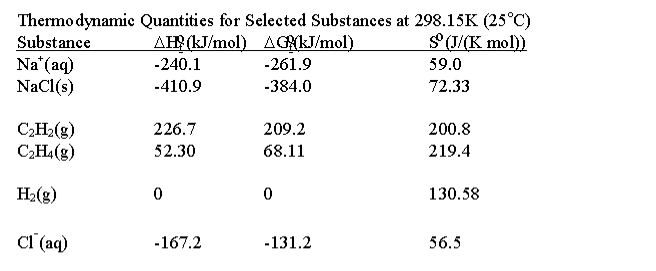
Another Thermo Question Socratic
18. Calculate the enthalpy of hydrogenation of C2H2 g to C2H4 g
C2H6 C2H4 H2. If the enthalpy change for this reaction is 137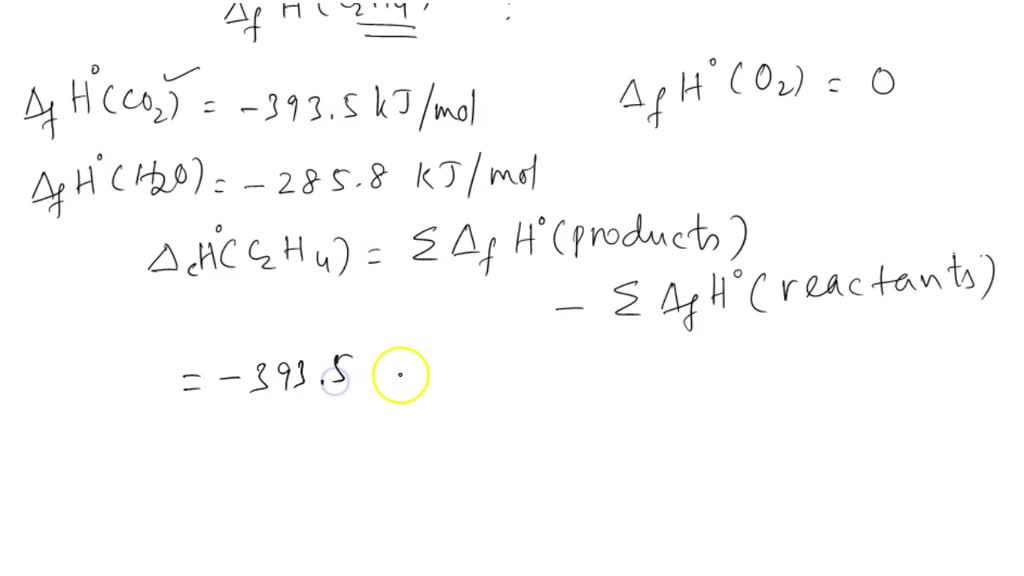
SOLVED The standard enthalpy of combustion of ethene gas C2H4 g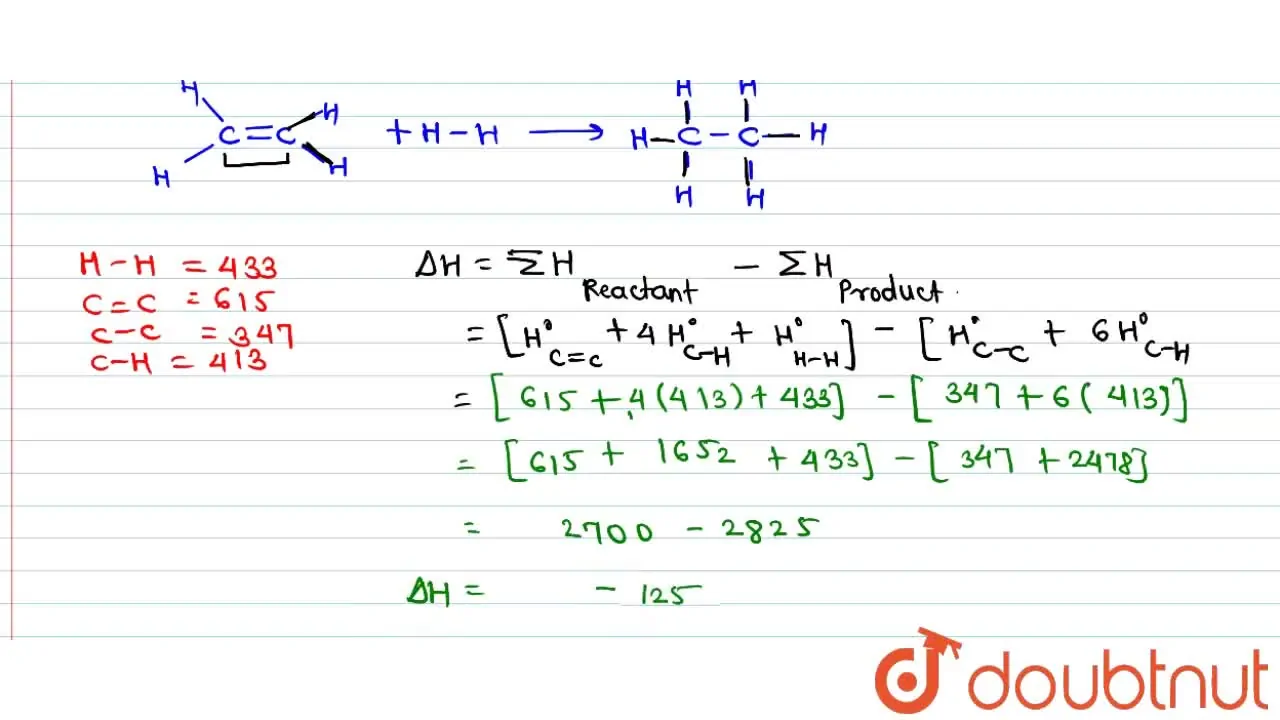
Marathi Calculate the enthalpy of hydrogenation of C2H4 C2H 4 g H
Given the following data Hf C2H4 12.5 kcal Heat of
Hf of c2H2 c2H4 c2H6 respectively are 226 56.the difference
24. Delta Hf C2H4 12.5 kcal Heat of atomization of c 171 kcal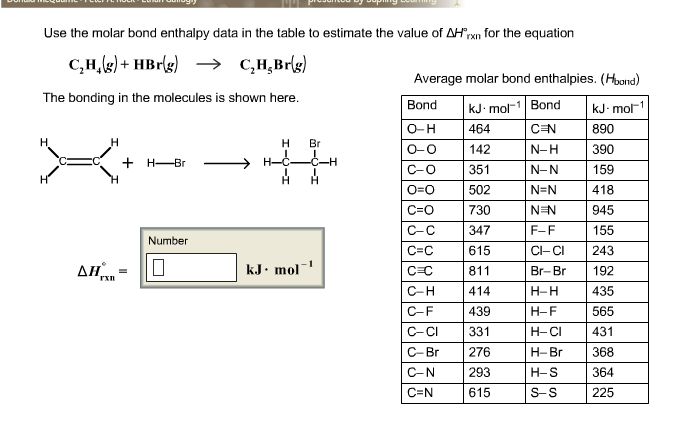
Solved Use the molar bond enthalpy data in the table to Chegg
SOLVED The enthalpies of combustion for ethene C2H4 ethane
ntif enthalpies of formation for c2h4 g co2 g and h2o l at
