
ethene molecule C2H4 Lewis dot cross electronic diagram sigma pi
C2h4 hot sale shared electrons
Share. Visit »
How many shared pairs of electrons are in the molecule C2H4 Quora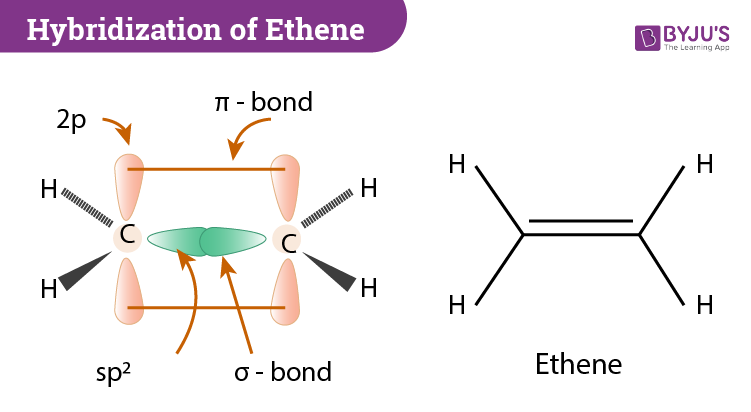
Hybridization of C2H4 Ethene Hybridization of Carbon in C2H4
Ethene with the molecular formula C 2 H 4 hasA. 5 covalent
Describe the bonding in ethylene C 2H 4 in terms of valence bond
Solved 3. a Draw the Lewis structure for ethylene C2H4 b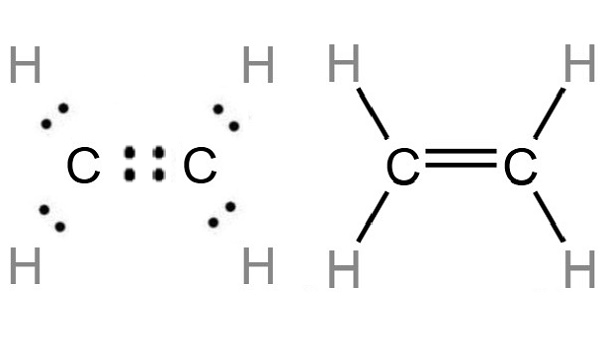
What is the Lewis structure of Ethylene Chemicalbook
Covalent bonding in an ethene molecule C2H4
Covalent bonding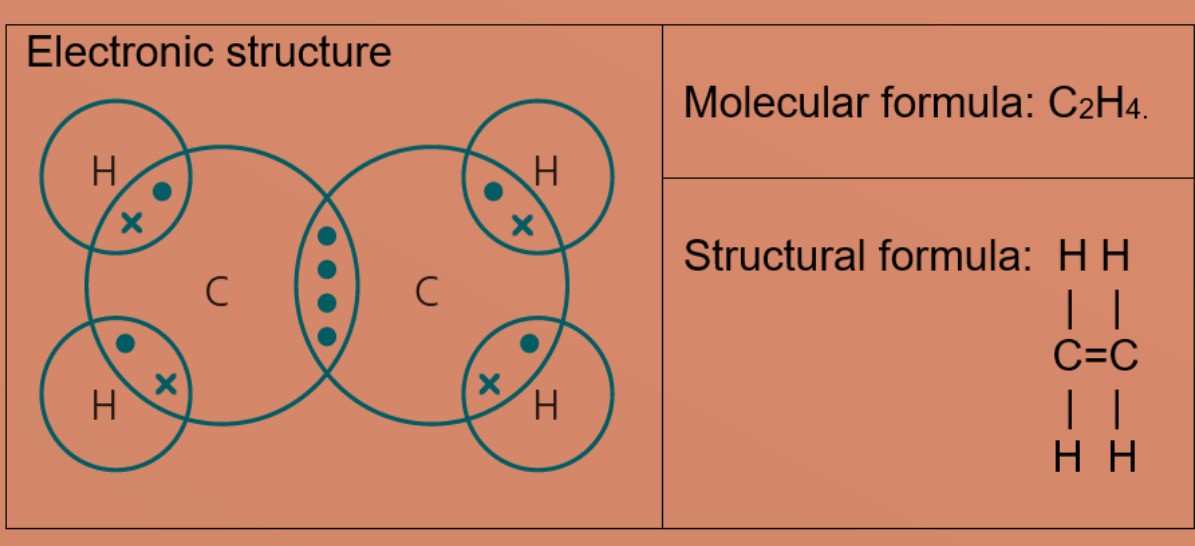
covalent bonding and molecules
The compound ethylene C2H4 is represented by this diagram. What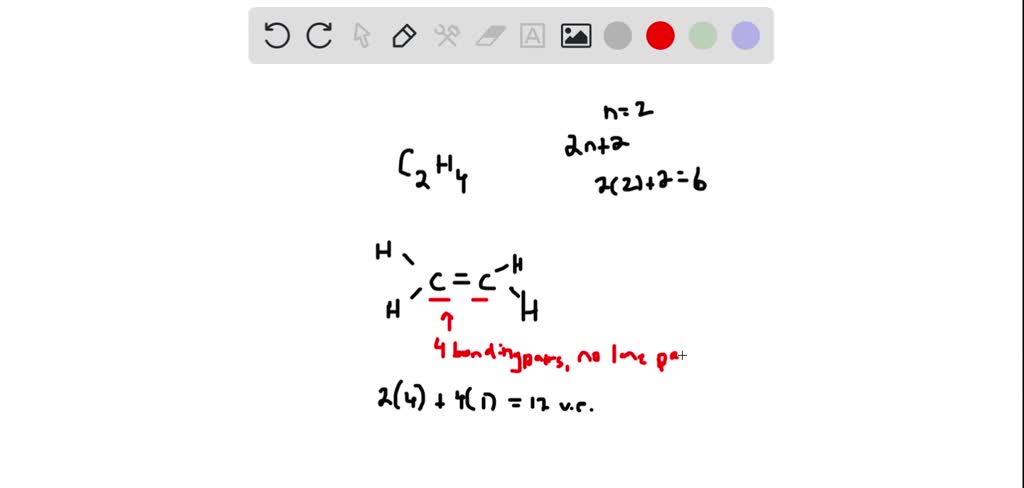
SOLVED Can someone help me find these answers C2H4 Number of
Is C2H4 Ethene or Ethylene Ionic or Covalent Molecular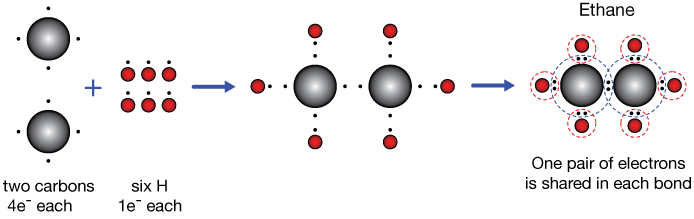
Covalent bonding
C2H4 Hybridization Ethylene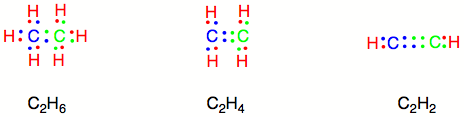
Life Sciences Cyberbridge
How many electron pairs are shared between carbon atoms in a
Lewis Structure for C2H4
C2H4 Lewis Structure Ethylene
What is the Lewis structure of Ethylene Chemicalbook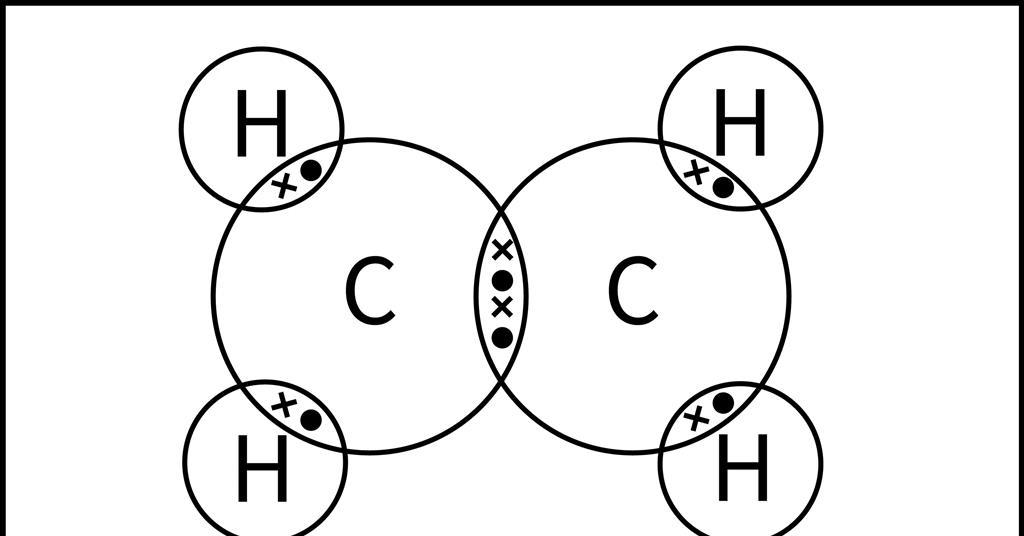
How to teach covalent bonding CPD RSC Education
C2H4 Lewis Dot Structure How to Draw the Lewis Structure for C2H4
How to Draw the Lewis Dot Structure for C2H4 Ethene YouTube
Draw the Lewis dot structure for C2H4 and a second structure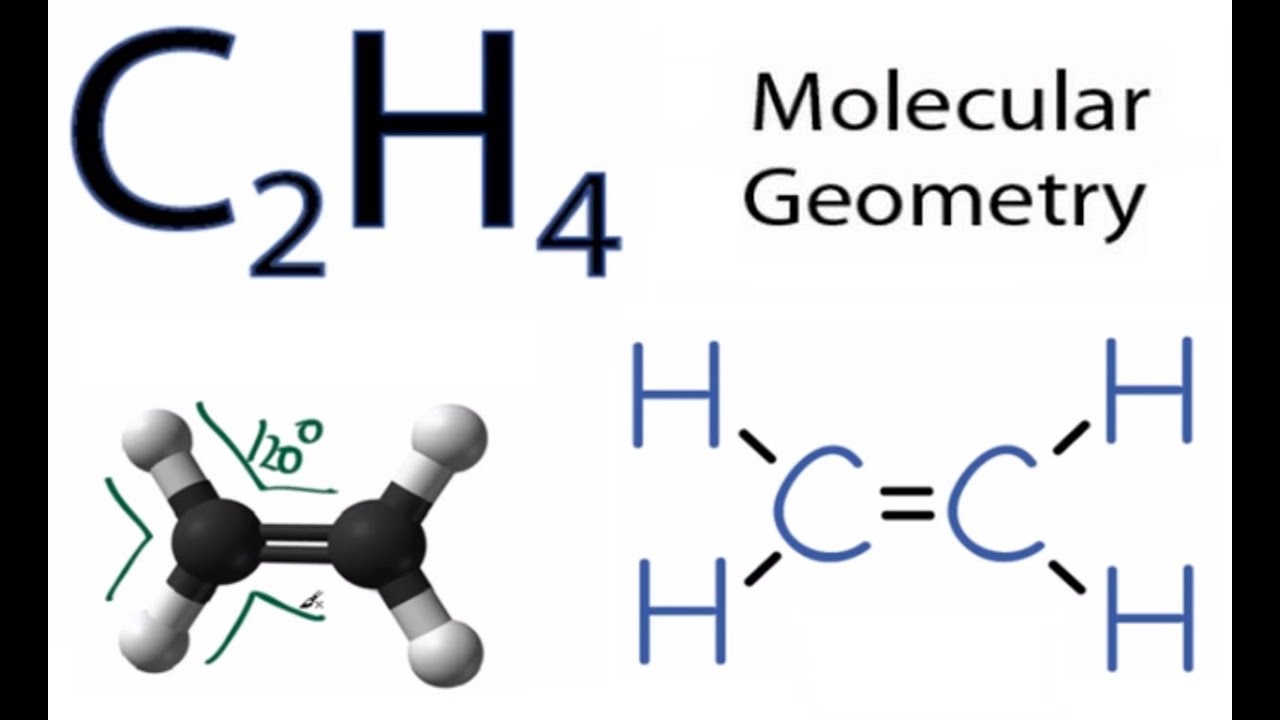
C2H4 Molecular Geometry Shape and Bond Angles
How many electron pairs are shared between carbon atoms in a
covalent bond
In the Lewis Dot structure of ethylene glycol would one hydrogen
Explain the formation of ethene molecule with the help of a
Draw the electron dot structure of ethene C2H4
Draw the Lewis structure of C2H4 and identify its geometry
How many electron pairs are shared between carbon atoms in a
Covalent Bonding Write down the information on these slides so
Write the electron dot structure of ethene molecule C 2H 4
Draw the Lewis dot structure for C2H4 and a second structure
Provide the following information for the compound C2H4. a. Lewis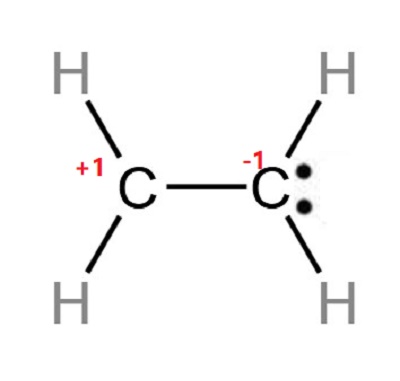
What is the Lewis structure of Ethylene Chemicalbook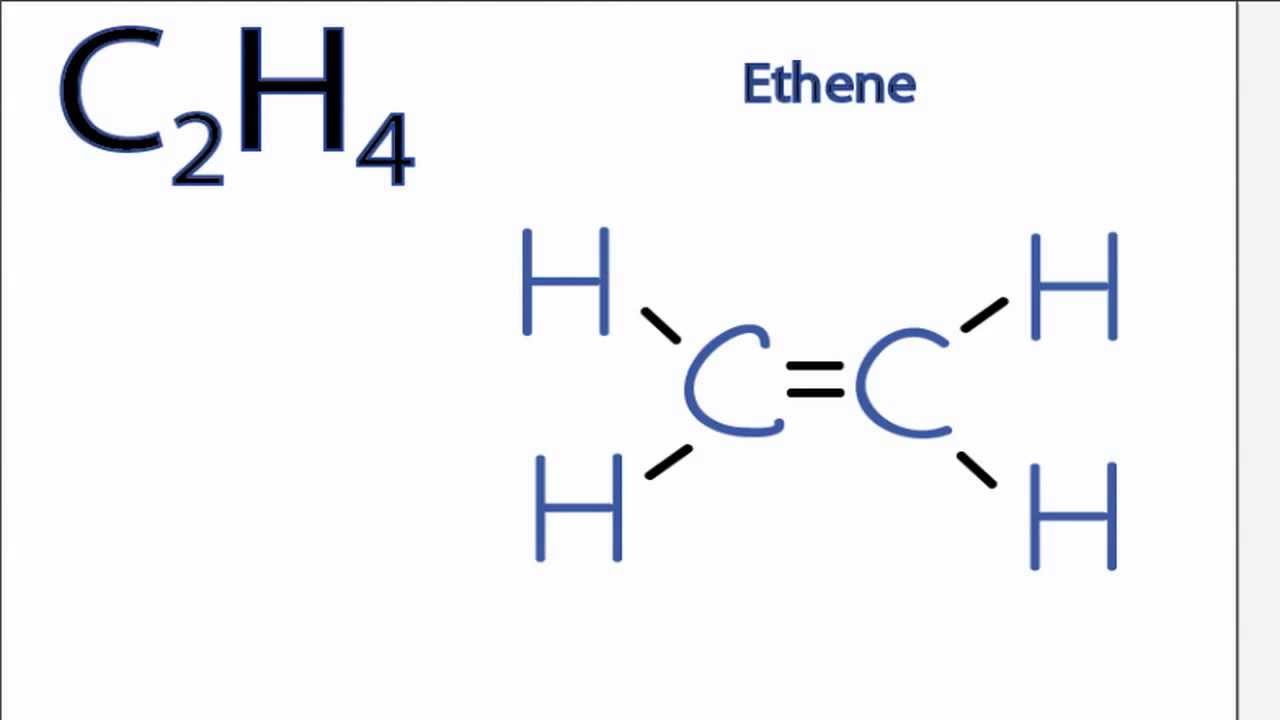
Is C2H4 Polar or Nonpolar
Draw Lewis structures for the following molecular formulas C2H4
How many electron pairs are shared between carbon atoms in a