
The enthalpy change for the following reaction is 137 kJ. Using
C2h4 h2 c2h6 hot sale using bond enthalpies
Share. Visit »
Solved H2 g C2H4 g C2H6 g BE C H 413 kJ BE H H
The average C H bond energy in CH4 is 415 kJ mol. Use the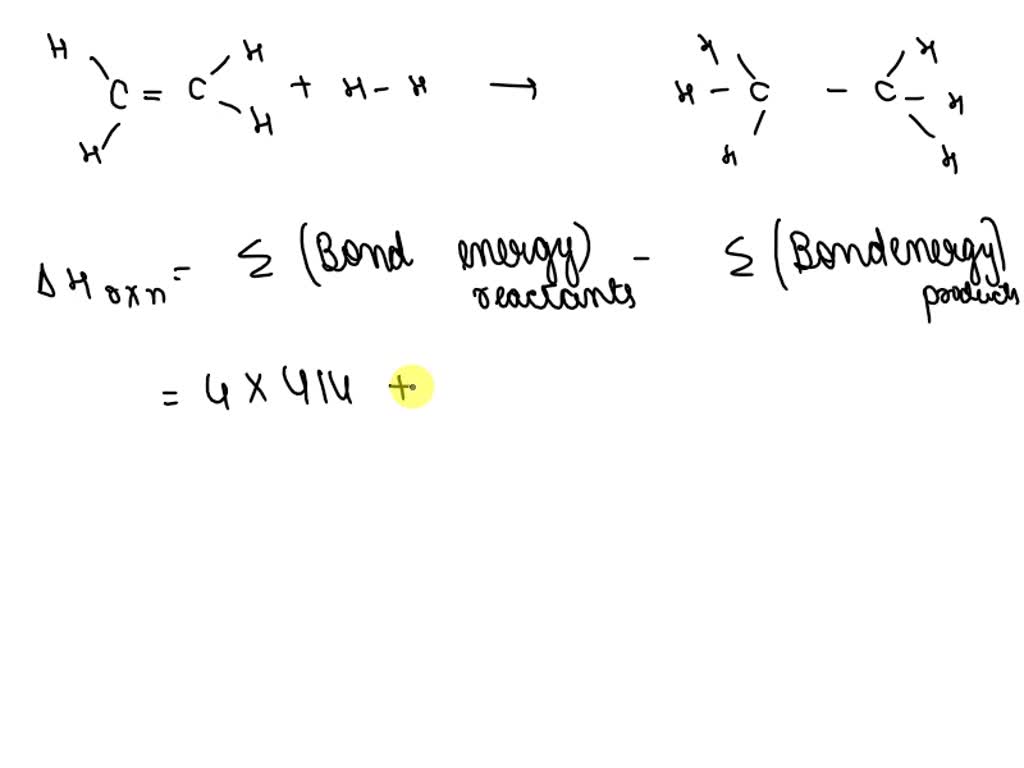
SOLVED Use the bond energies provided to estimate Hrxn for the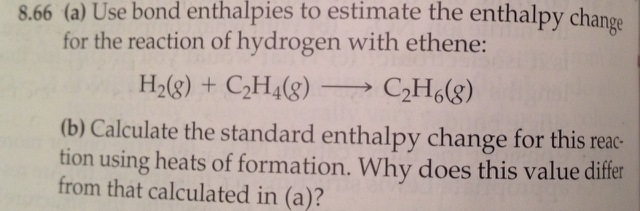
Solved 8.66 a Use bond enthalpies to estimate the enthalpy
5 4 0. 112L From the following thermochemical equations C2H4 H2
Solved Estimate the enthalpy change for the reaction C2H4 g
SOLVED H2 g C2H4 g C2H6 g BE C H 413 kJ BE H H 436
For the reaction C2H4 H2 C2H6 E 30 kcal YouTube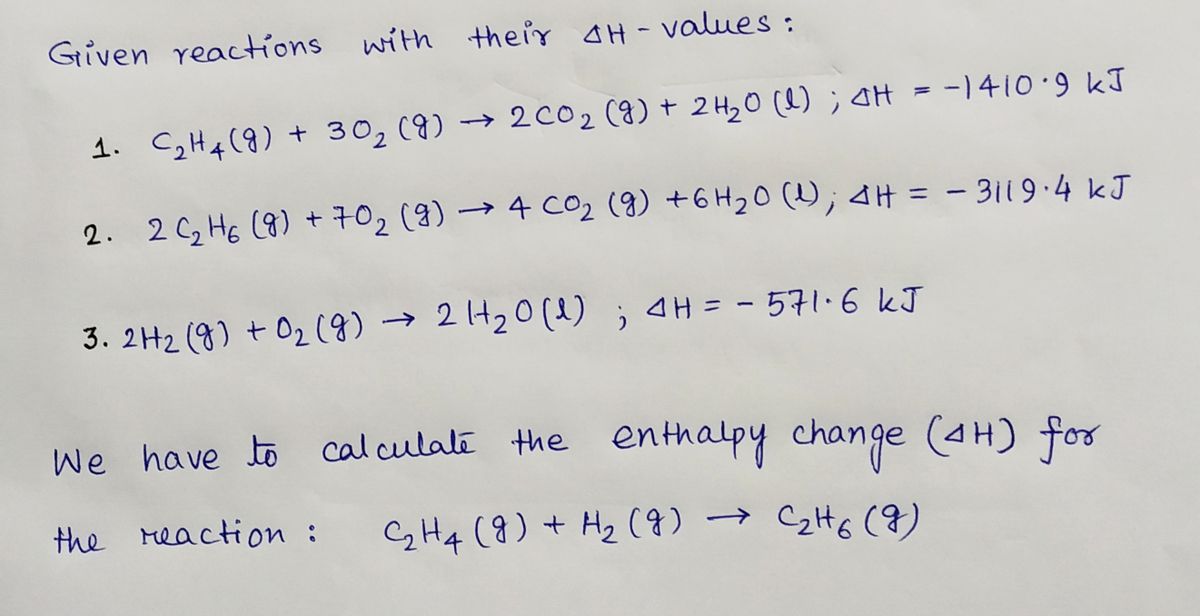
Answered Calculate the enthalpy change for the bartleby
Answered 2. Calculate the enthalpy change for bartleby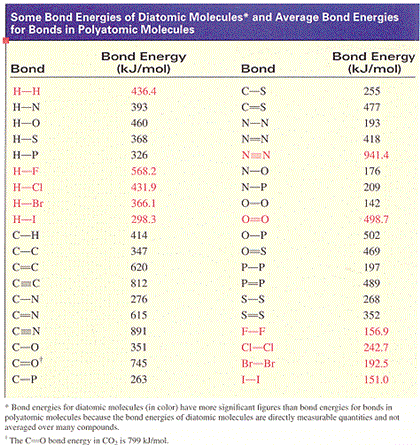
Solved H2 g C2H4 g C2H6 g a Estimate the enthalpy of Chegg
Use standard enthalpies of formation to calculate H rxn for eac
58. The enthalpy change the reaction H g C H. g C H g
Solved Using the values of bond energy from the table above
IN THE SOLUTION WHY IS THE UNDERLINED STEP USED H reactants H
Hf C2H4 12.5 kCal Heat of atomisation of C 171 kCal Bond
Use bond energies to confirm that the complete combustion of
SOLVED Draw Lewis structures for reactants and products. Estimate
18. Calculate the enthalpy of hydrogenation of C2H2 g to C2H4 g H2(g) → C2H6(g) ΔH = = -125 kJ mol-1.jpg)
International Baccalaureate Chemistry ppt download
The enthalpy change for the following reaction is 137 kJ. Using
Solved Use the bond energies provided to estimate AH rxn for H2(g) → C2H6(g).jpg)
1.2.6 Bond Enthalpies. ppt download
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g
Solved Use bond enthalpies above to estimate the enthalpy Chegg
Solved Use bond enthalpies to estimate the enthalpy change Chegg
a Volumetric fractions of H2 CO O2 C2H4 and C2H6 using the
Solved Question H2 g C2H4 g C2H6 8 BE C H 413
Solved From the data below calculate the approximate Chegg
For the reaction C2H4 H2 C2H6 E 30 kcal
58. The enthalpy change the reaction H g C H. g C H g
a CH4 covalent bond and bond hybridized orbital b C2H6
For the reaction C2H4 H2 C2H6 E 30 kcal YouTube
Answered 2. a The average C H bond energy in bartleby
The heat of combustion of C2H4 C2H6 and H2 are 1409.5 kJ 1558.3kJ a
Solved H2 g C2H4 g C2H6 g BE C H 413 kJ BE H H
C2H6 C2H4 H2. If the enthalpy change for this reaction is 137
The enthalpy change for the reaction H2 C2H4 C2H6 is The
7.65c Calculate the enthalpy change from bond energies C2H4 g 3O2 g 2CO2 g 2H2O g


