
Orbital Overlap Diagram for C2H4 Ethene acetylene double bond
Pi and sigma hot sale bonds in c2h4
Share. Visit »
a Sigma Single bond hybridization for ethylene b pi Single
Indicate the sigma and pi bonds in the following molecules.C 6H 6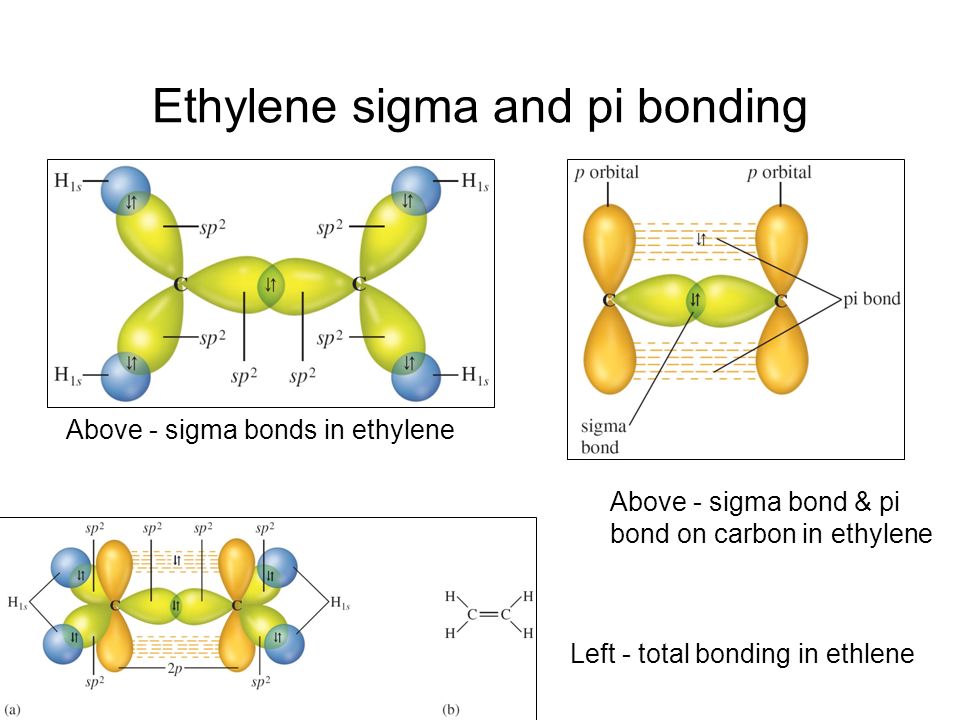
Localized e model and hybrid orbitals sigma and pi
Draw Lewis structures for the following molecular formulas C2H4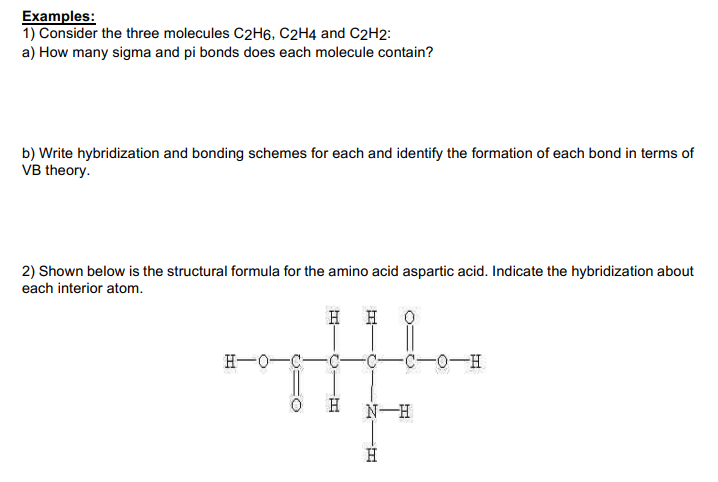
Solved Examples 1 Consider the three molecules C2H6 C2H4
Sigma And Pi Bonds Covalent Bonds MCAT Content
How many sigma bonds and pi bonds are present in C2H4 Ethene
13.2. Molecular orbitals for ethene Organic Chemistry II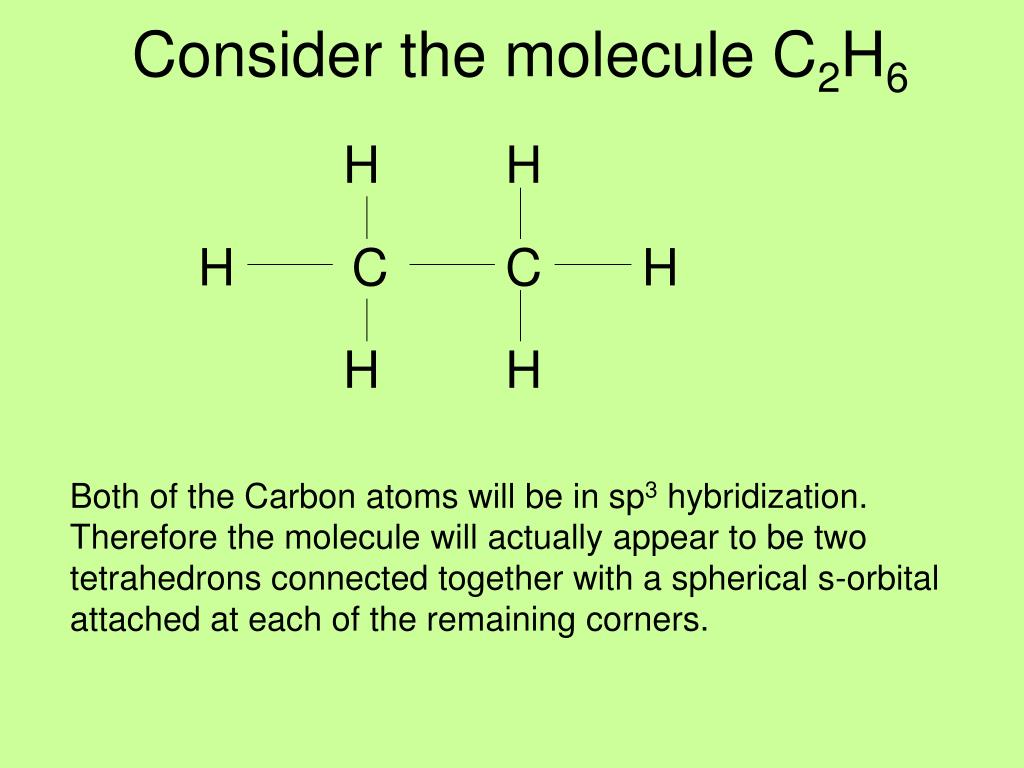
PPT Chemical Bonding PowerPoint Presentation free download ID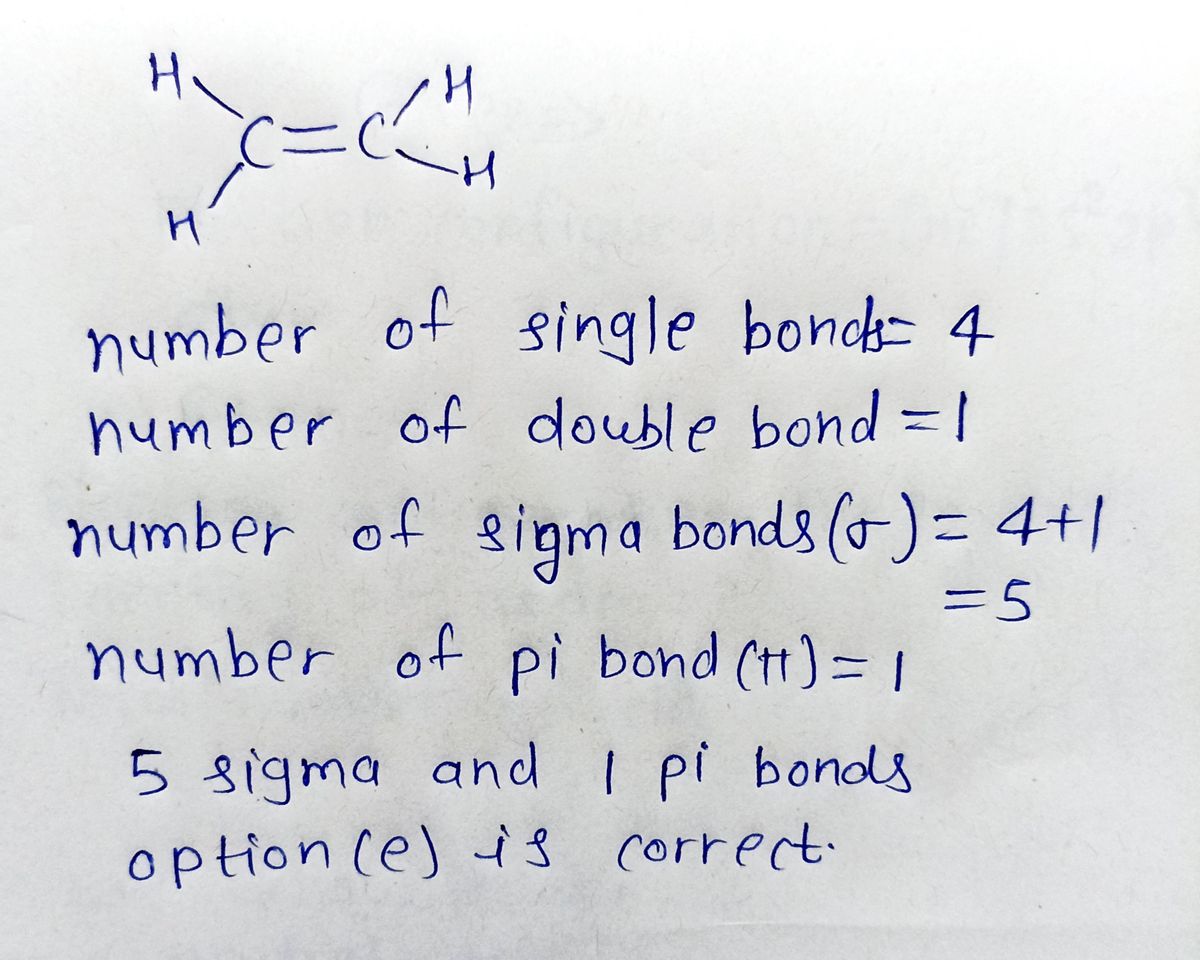
Answered Consider the ethylene molecule C2H4. bartleby
How many sigma and pi bonds are there in the following molecules
Solved How many sigma and pi bonds are present in C2H4 a.5
How many sigma bonds are present in ethane Quora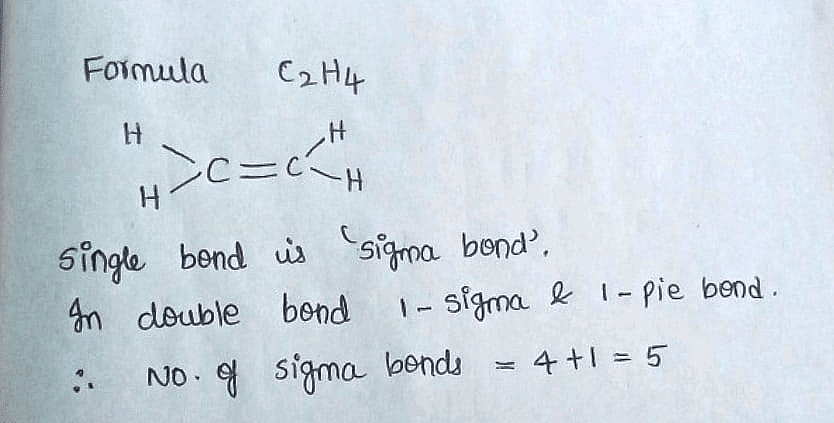
What do you think is the number of sigma bonds in an ethene
Sigma And Pi Bonds Covalent Bonds MCAT Content
Sigma and Pi Bonds Read Chemistry CK 12 Foundation.png)
Explain the formation of ethene molecule c2h4 by double bond Also
Solved Question 1 1 point HI Shown above are the carbon Chegg
The outer orbitals of C an Ethene molecule can be considered to be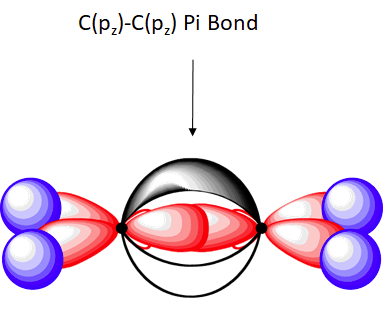
1.8 sp Hybrid Orbitals and the Structure of Ethylene Chemistry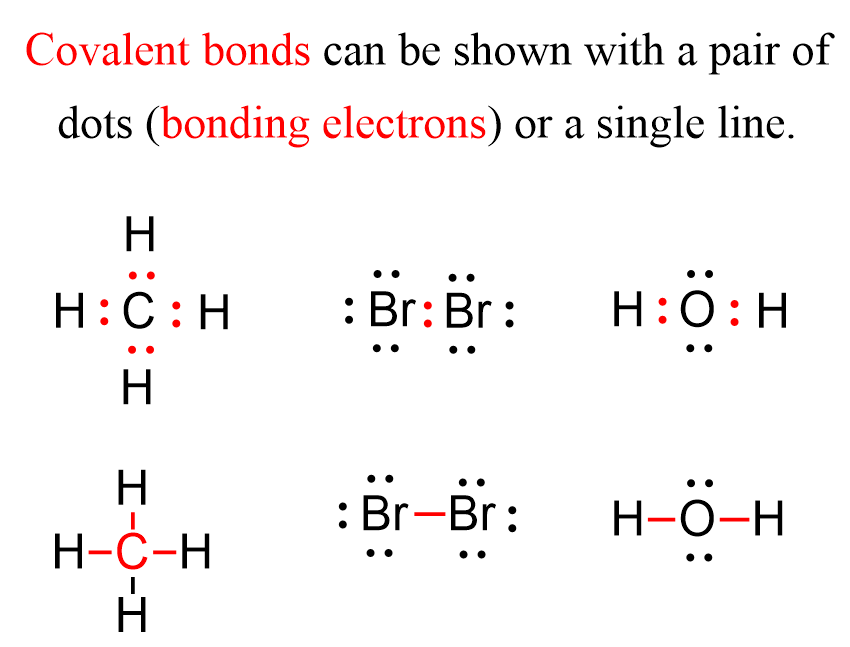
Sigma and Pi Bonds Chemistry Steps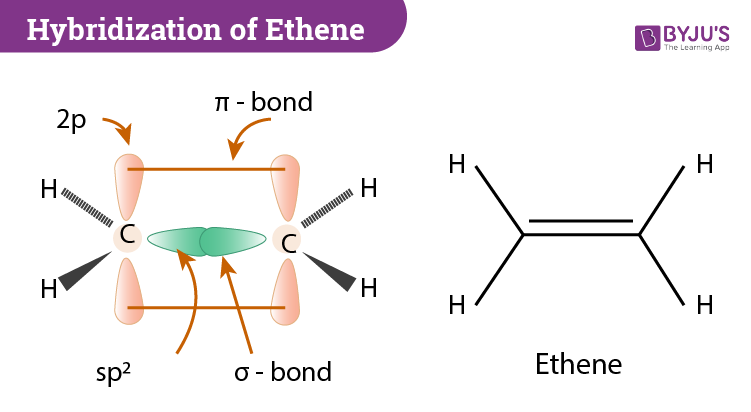
Hybridization of C2H4 Ethene Hybridization of Carbon in C2H4
Answered Consider the ethylene molecule C2H4. bartleby
What is the total number of sigma and pi bonds in C2H4 Ethene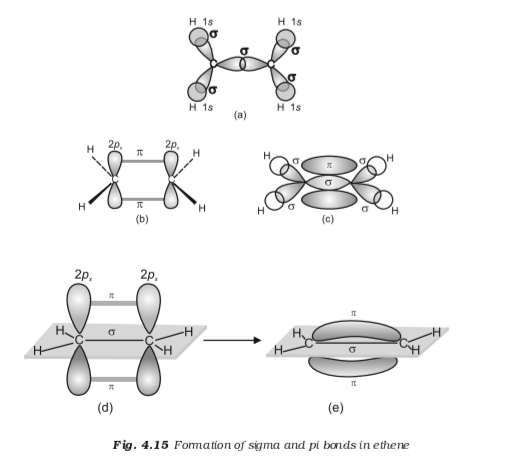
C2H4 Hybridization
How many sigma bond and pi bond are there in C2H4 Explain with
How many sigma bonds are formed in ethene Quora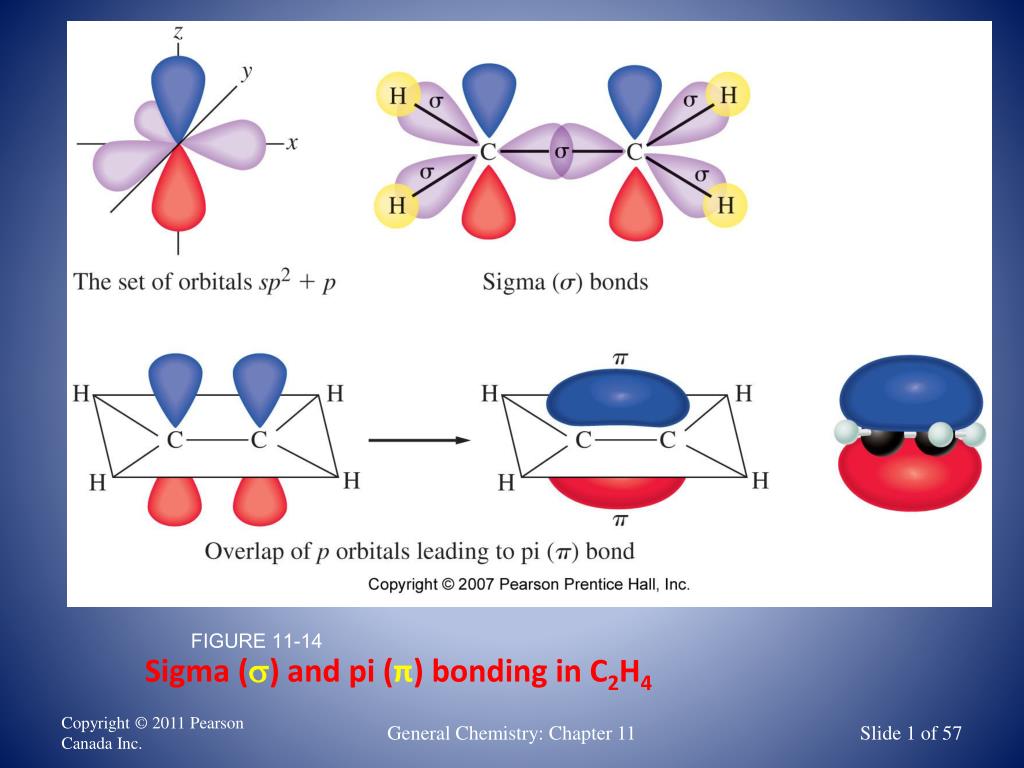
PPT Sigma s and pi bonding in C 2 H 4 PowerPoint
What are the total number of sigma and pi bonds in the following
What is the total number of sigma and pi bonds in the following molecules a C2H2 b C2H4
Sigma and Pi Bonds Chemistry Steps
a CH4 covalent bond and bond hybridized orbital b C2H6
Solved Consider this molecule ethene a Draw a sketch of the
Solved Describe the bonding in ethylene C2H4 in terms of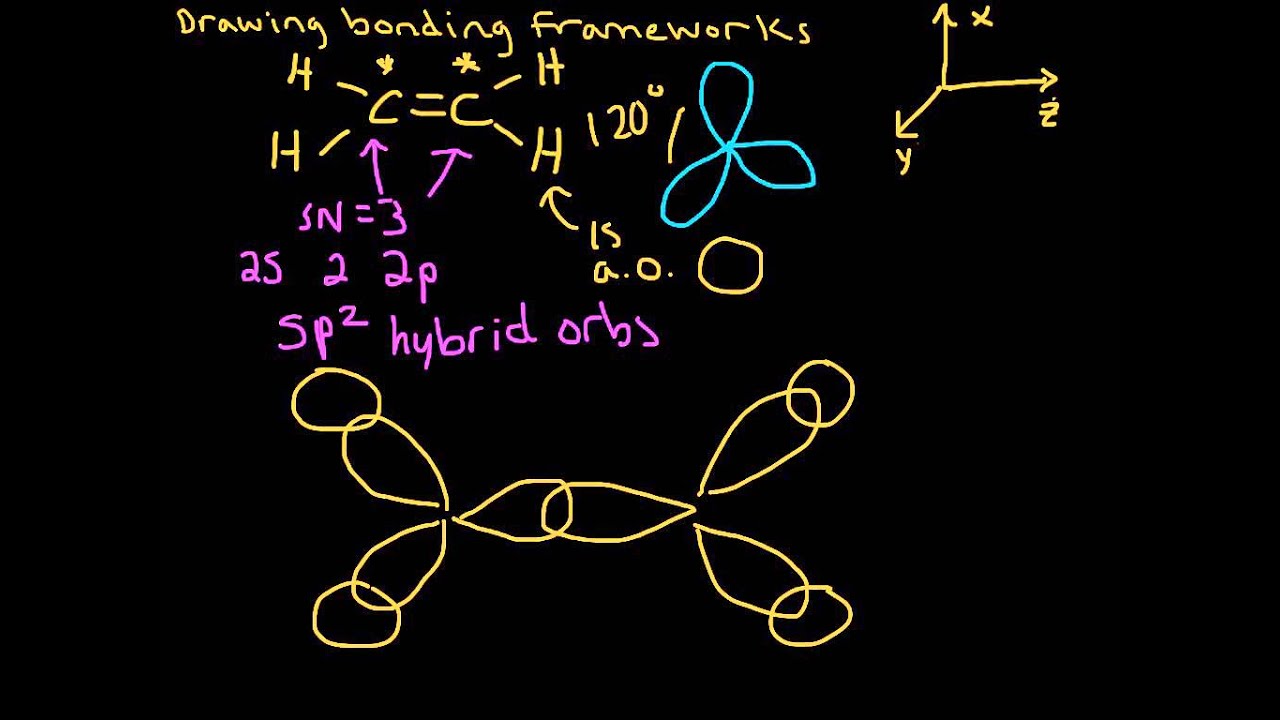
Sigma and Pi Bonding in Ethene C2H4 Hybridization
Types of Hybridization and Hybrid Orbitals
indicate the number of pi bonds in C2H4. Homework.Study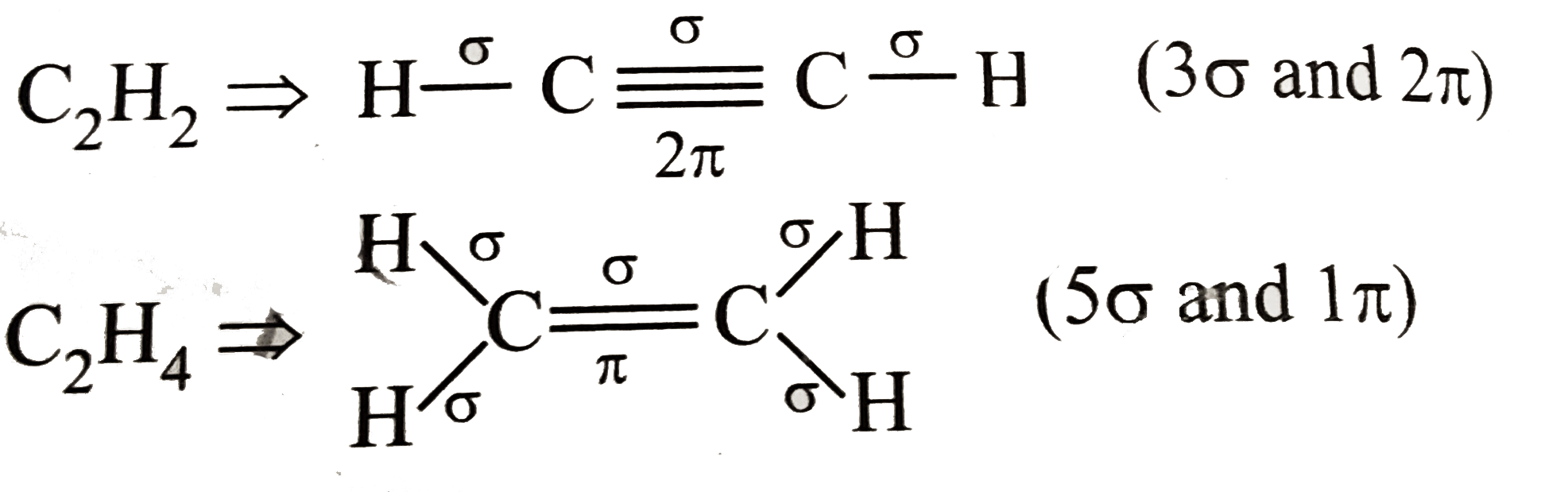
what is the total number of sigma and pi bonds in the following molecu
How many sigma bonds and pi bonds are present in C2H4 Ethene
