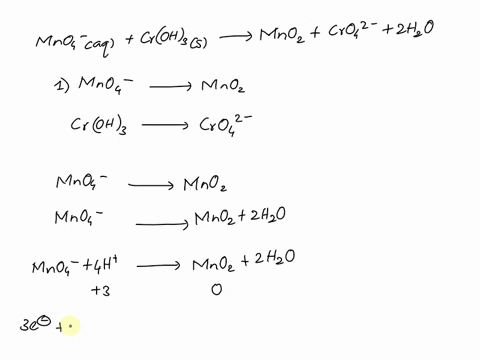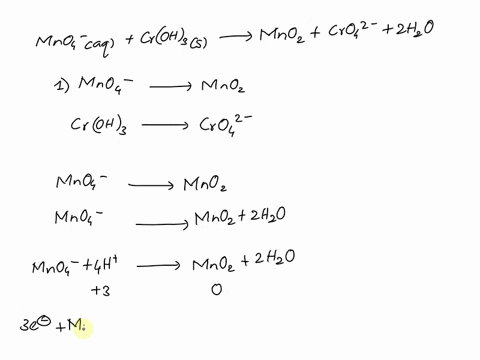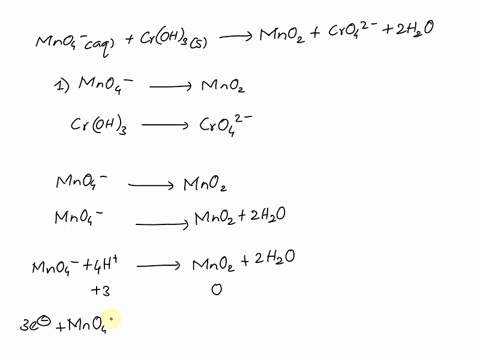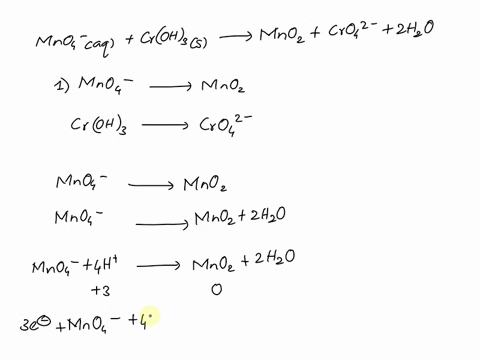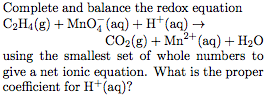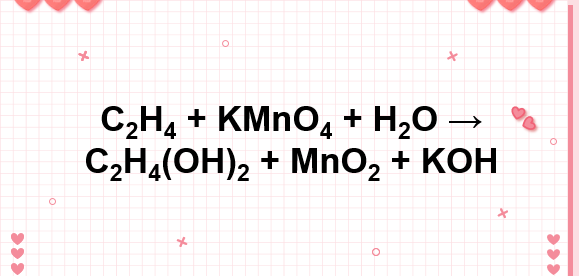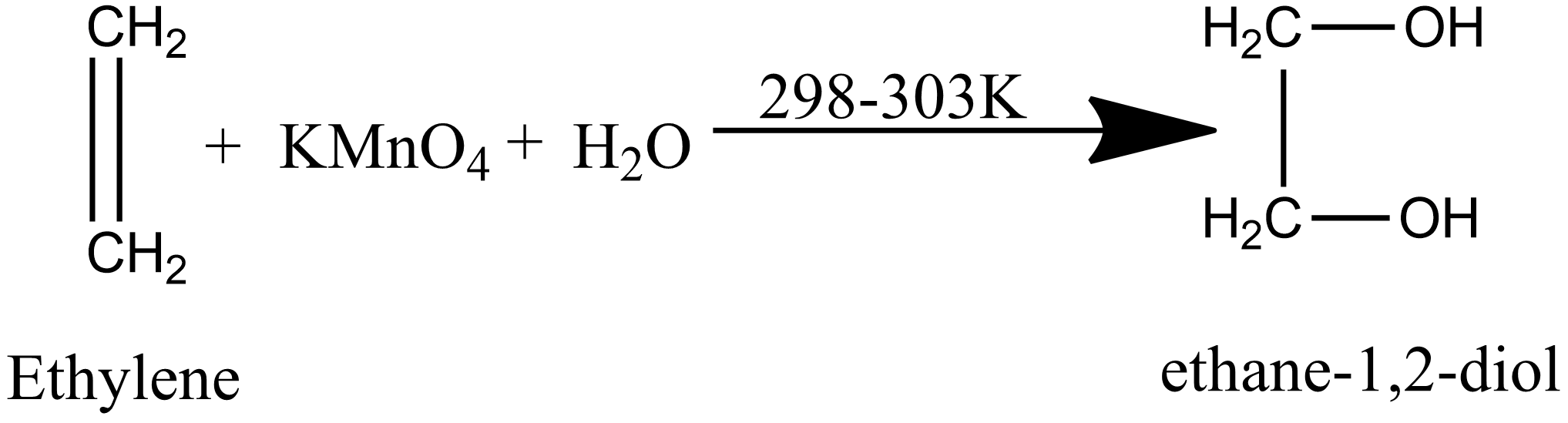Ethylene oxidation reaction by potassium permanganate based
C2h4 mno4 hot sale
Share.
Visit »
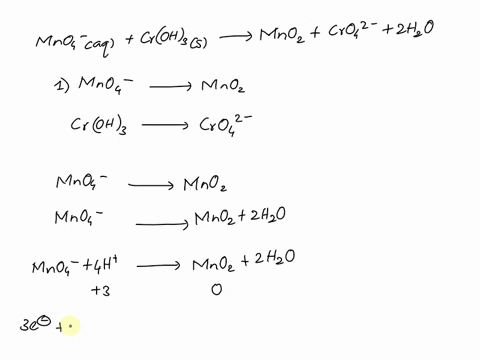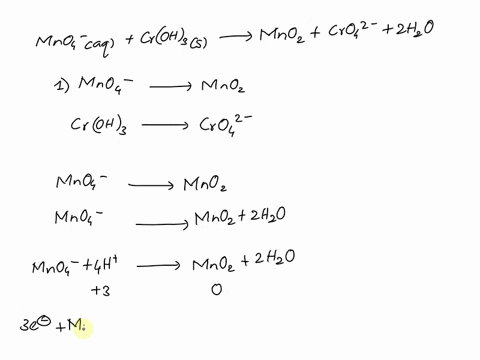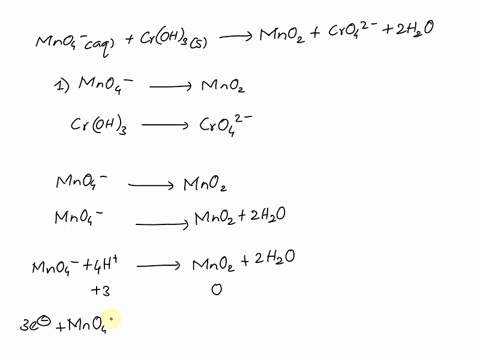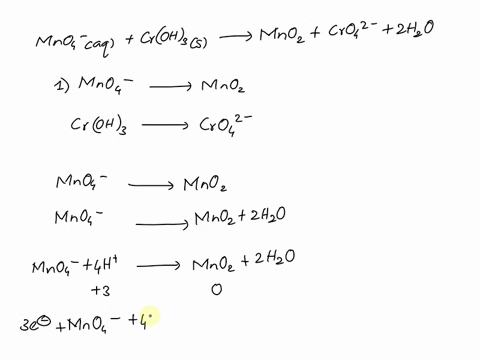
SOLVED Balance this reaction in acid C2H4 g MnO4 aq 
C2H4 KMnO4 H2SO4 CO2 K2SO4 MnSO4 H2O Balanced 
When ethylene is passed through alkaline KMnO 4 solution we get 
Acetylene and ethylene react with alkaline KMnO 4 to give 
How exactly is ethene oxidized by potassium manganate VII Quora 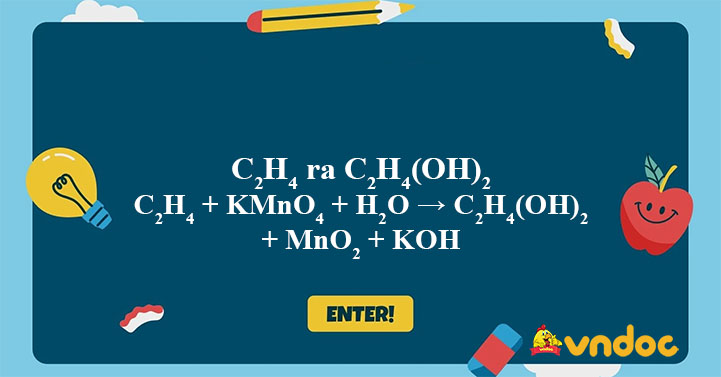
C2H4 KMnO4 H2O C2H4 OH 2 MnO2 KOH C2H4 ra C2H4 OH 2 
MnO4 C2O4 2 Mn 2 CO2 
Solved Balance this reaction in base C2H4 g MnO4 aq 
A 2Cl Cl2 2e x3Cr2O7 2 14H.pdf 
MnO4 C2O42 H Mn2 CO2 Balance the following 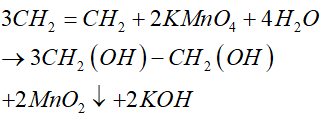
D n t t kh C2H4 v o dung d ch KMnO4 hi n t ng quan s t c l 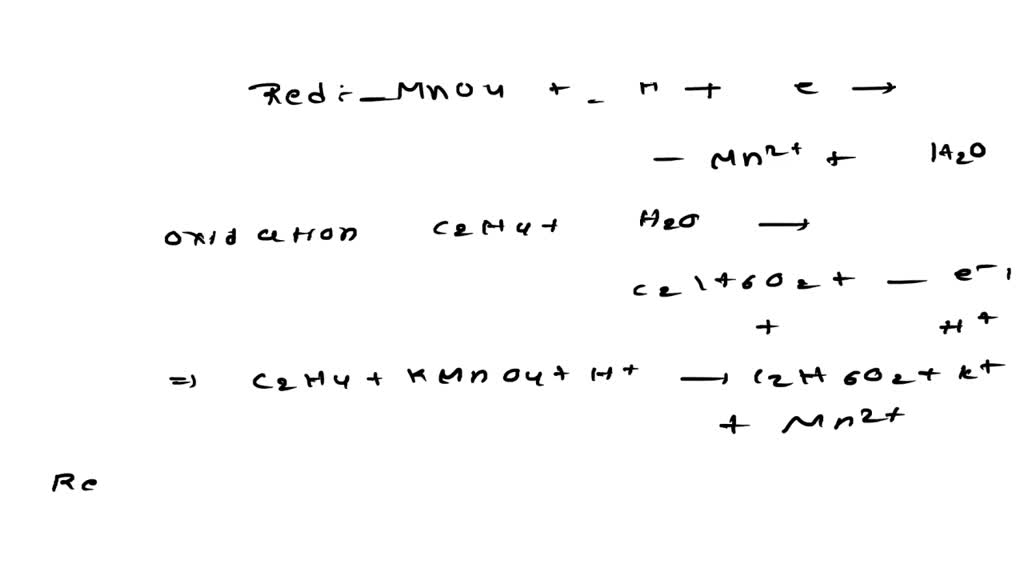
SOLVED The reaction of an alkene with acidic potassium 
Solved This question has multiple parts. Work all the parts 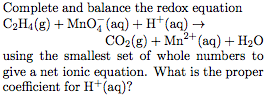
Solved Complete and balance the redox equation C2H4 g Chegg 
What is BEfresh Befresh 
SOLVED Balance this reaction in acid C2H4 g MnO4 aq 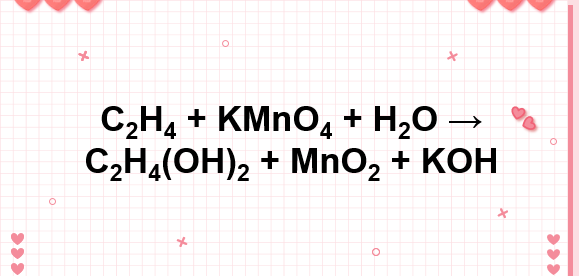
C2H4 KMnO4 H2O C2H4 OH 2 MnO2 KOH C2H4 ra C2H4 OH 2 
Mail.ru . . 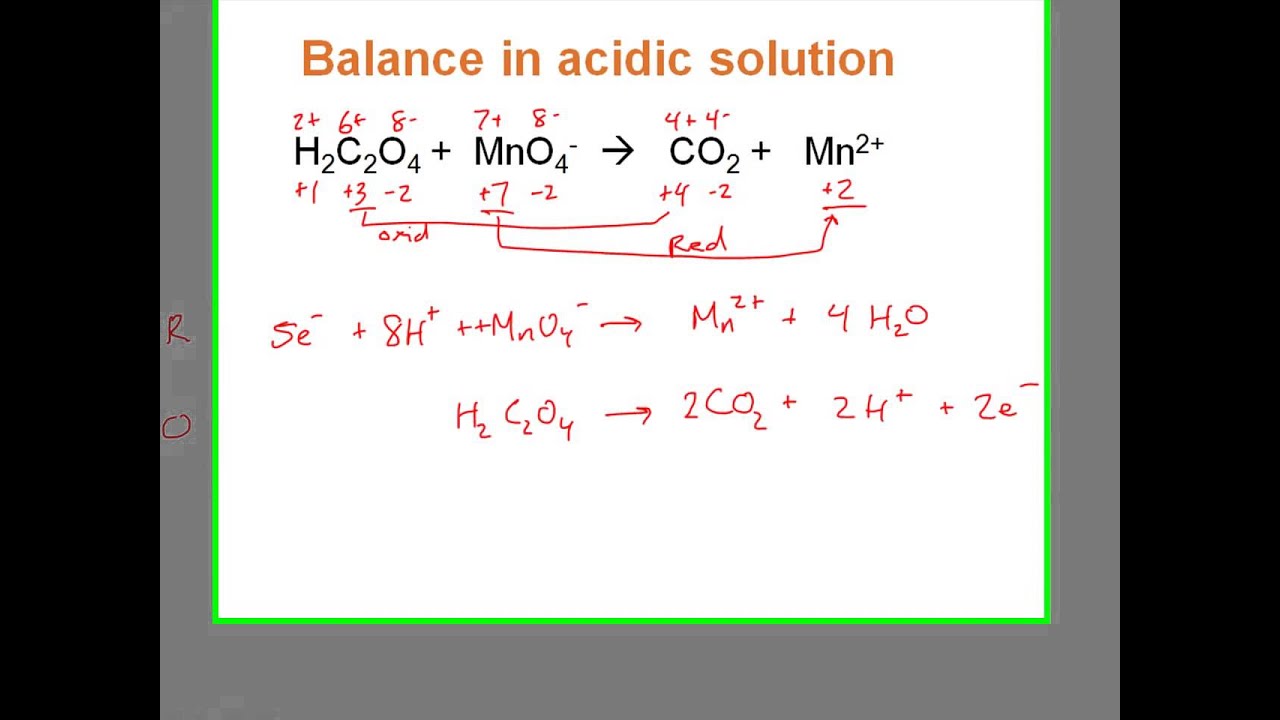
Balancing Redox Equation with MnO4 
A guide for A level students KNOCKHARDY PUBLISHING ppt download 
Solved Ethylene reacts with alkaline KMnO4 to form 
Metode PBO Penyetaraan 16 MnO4 C2H4 H2O MnO2 OH 
c2h4 kmno4 h2o ch2oh ch2oh mno2 koh 
KMnO4 concentration in C2H4 scrubber sachets during blueberry 
Solved Balance the folowing redox reactions under acidic Chegg 
Metode Transfer Hidrogen H Reaksi Redoks MnO4 C2H4 MnO2 
Balancing Redox Equation with MnO4 YouTube 
C2H4 KMnO4 CH2 CH2 KMnO4 H2O OH CH2 CH2 OH MnO2 
Schematic representation of an C2H4 scavenger sachet used in 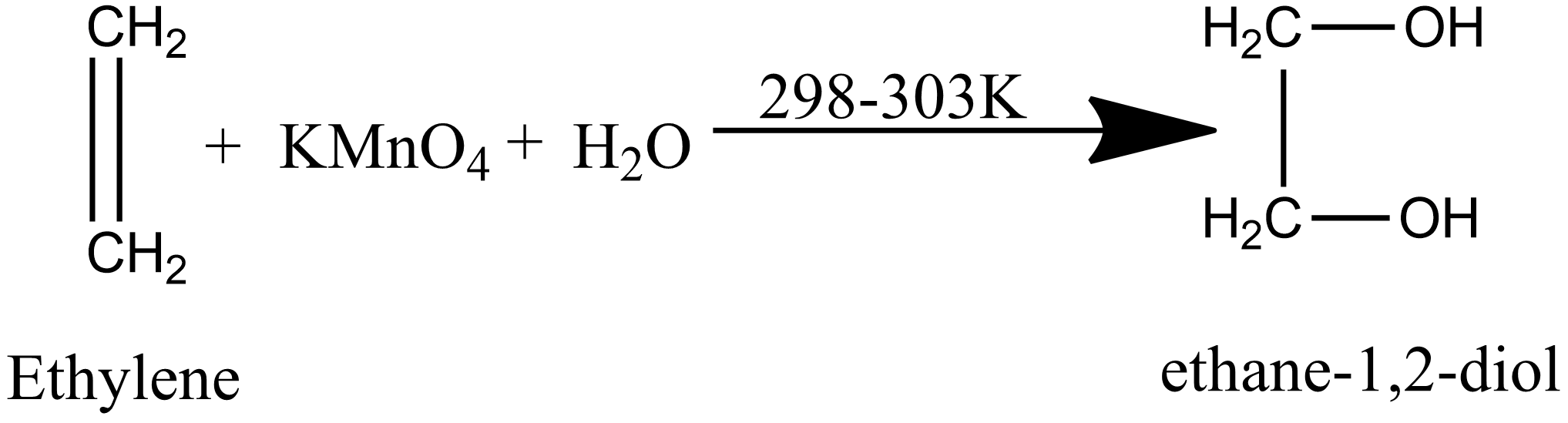
Ethylene on reaction with alkaline Kmn O 4 gives A. GlycerolB 
What happens when Ethylene is treated with alkaline KMnO 4 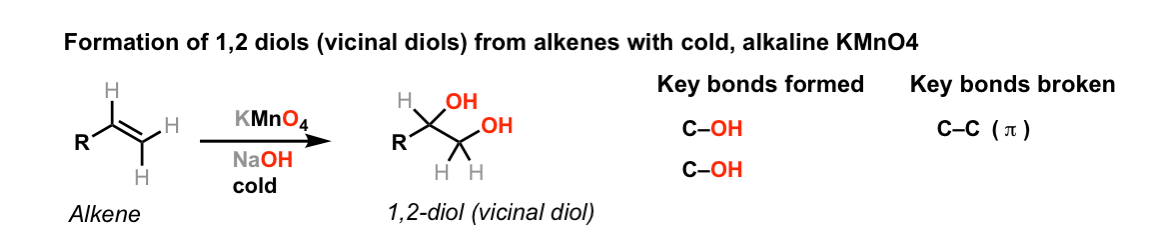
Dihydroxylation of alkenes with cold dilute KMnO4 to give vicinal 
Changes in C2H4 uptake capacity a and KMnO4 concentration b 
C2H4 KMnO4 MnO2 C2H6O2 KOHNapisz r wnania redoks 
Solved 15. Balancing in base 1 points Balance this Chegg 
Solved Balance the following redox reaction using the Chegg 
What happens when ethylene gas is passed through a solution of 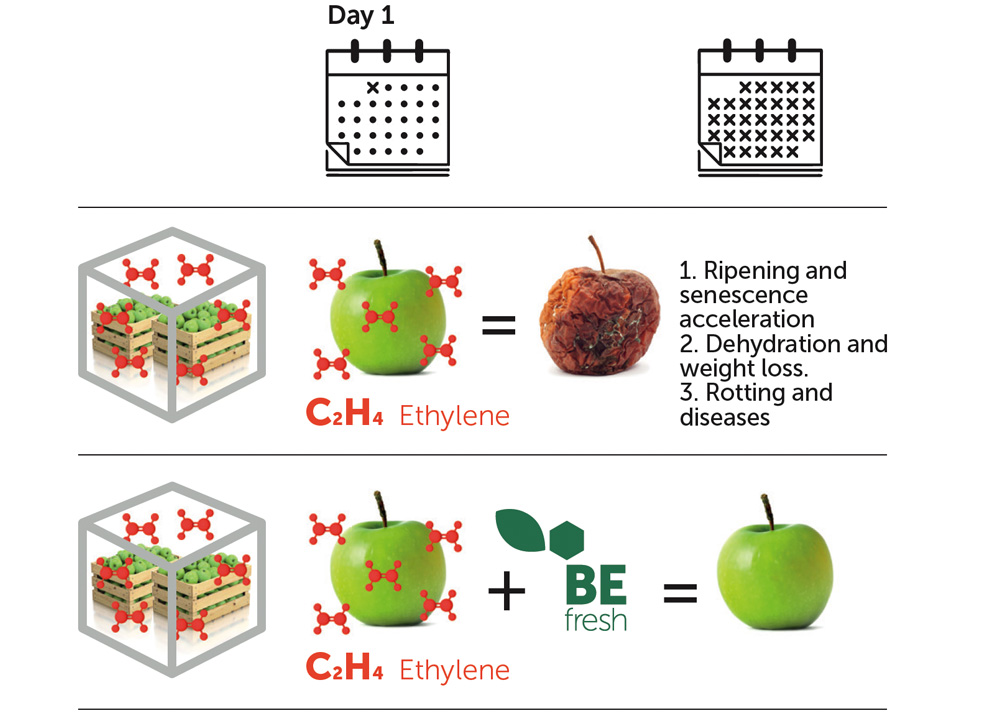
What is BEfresh Befresh 
Setarakan reaksi redoks berikut dengan metode bila