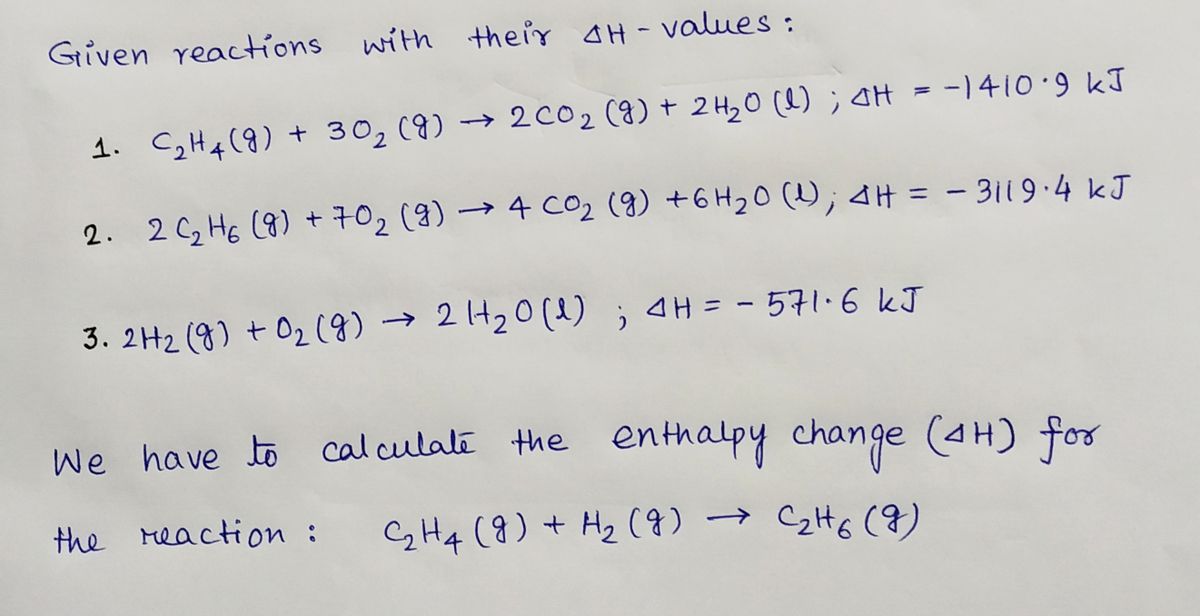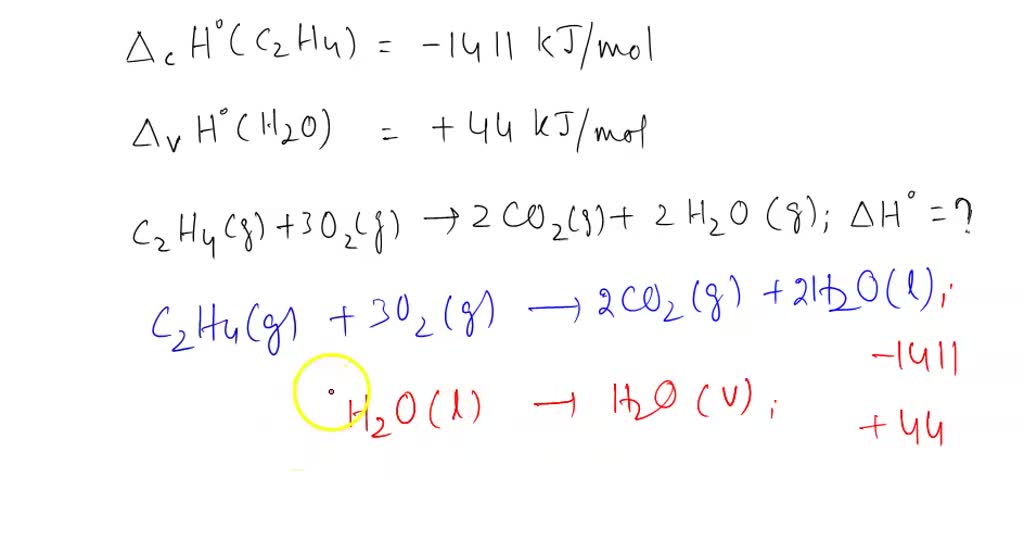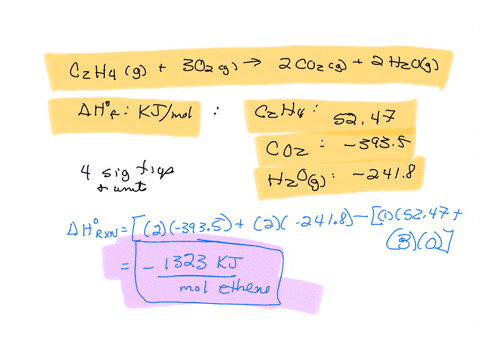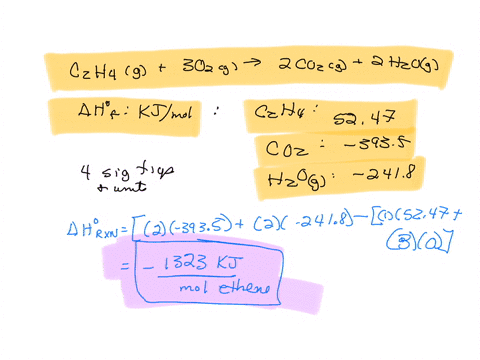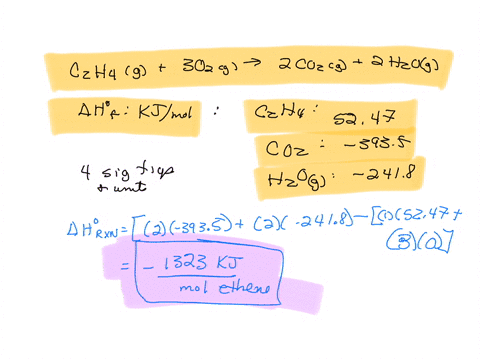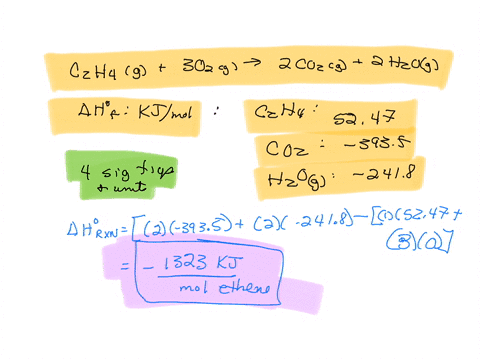Answered For the combustion reaction of ethylene bartleby
C2h4 kj/mol hot sale
Share.
Visit »

ntif enthalpies of formation for c2h4 g co2 g and h2o l at 
Assignment 8 Chemistry 30 Module 1 Lesson 5 1 Assignment 
ANSWERED CO2 g 393 5 kJ mol H O 1 285 8 kJ mol AH kJ mol An error 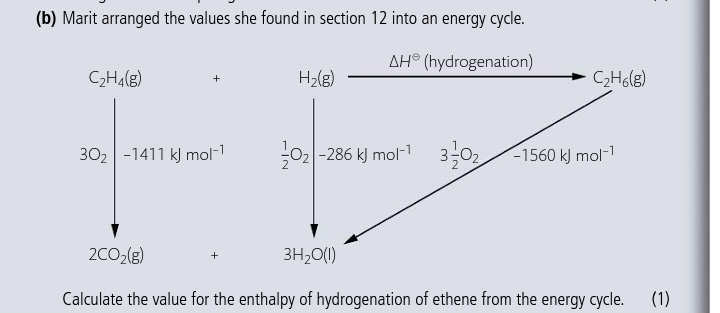
Enthalpy changes thermochemistry Practice Question Solving 
Be sure to answer all parts. Calculate the heats of combustion for 
Q If enthalpies of formation for C2H4 g CO2 g and H2O l at 25 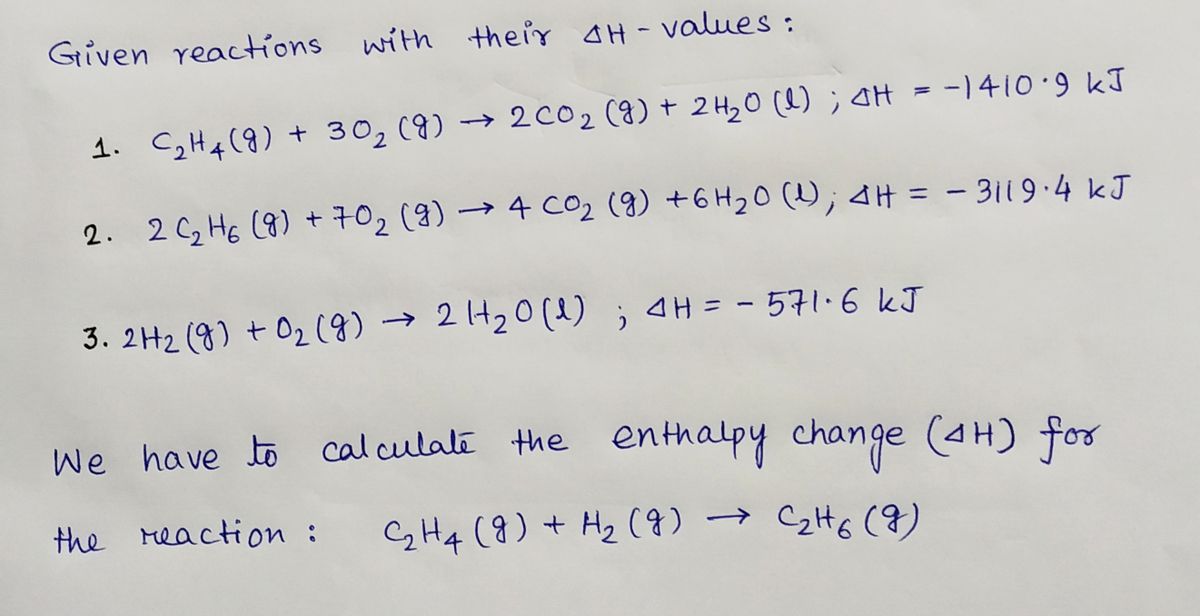
Answered Calculate the enthalpy change for the bartleby 
Solved Sample Question 8 What is the standard enthalpy of Chegg 
rolls Which reaction shows that the enthalpy of formation of C2H4 
In C2H4 formation of C C and C C is 590 kJ mole and 331 
Chemistry 4.4 Hess s law Flashcards Quizlet 
The enthalpy of formation for C 2 H 4 g CO 2 g and H 2 O l at 25 C and 1 atm. Pressure be 52 394 and 286 kJ mol 1 respectively. The enthalpy of combustion of 
0 22.0 HDI c 23.0 kJ mol d 92.0 kJ mol 19 Given the 
ASSIGNED READINGS 
Solved For the reaction C2H4 g H2 g C2H6 g H is 
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g 
Solved 2. The standard enthalpy of combustion of ethylene Chegg 
Pls help with this question r ALevelChemistry 
Answered The standard enthalpy of combustion of bartleby 
Alberta Inquiry into Chemistry February 12 2007 Page 1 of 16 Unit 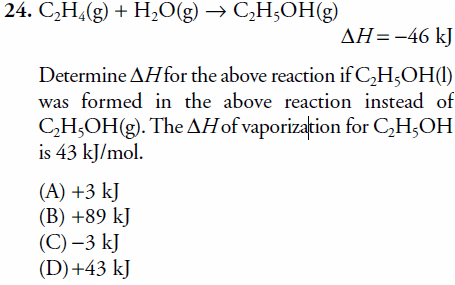
Understanding Hess Law C2H4 H2O C2H5OH 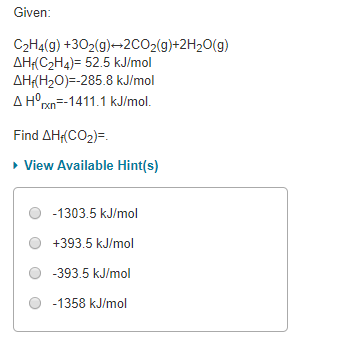
Solved Given C2H4 g 302 g 2CO2 g 2H20 g AH C2H4 52.5 
OneClass The combustion of ethane C2H4 occurs via the reaction 
Ethanol is manufactured in industry by the hydration of ethylene 
In C 2 H 4 energies of formation of C C and C C are 145 kJ mol a 
a Single component adsorption desorption isotherms of C2H6 and 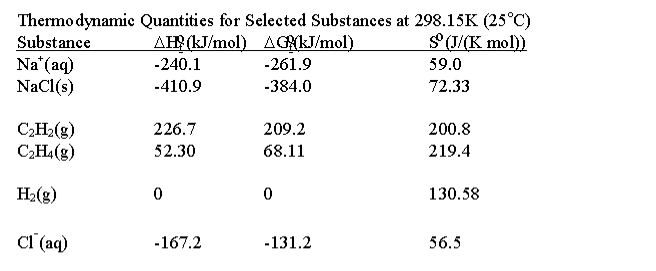
Another Thermo Question Socratic 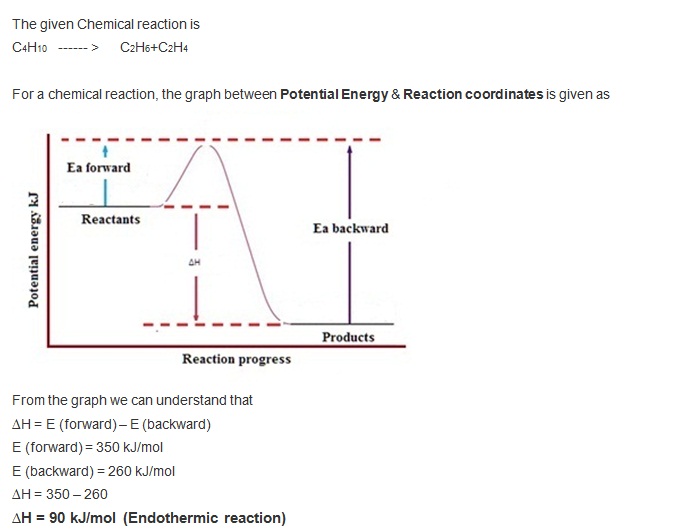
Solved the reaction C4H10 C2H6 C2H4 has an activation energy 
Be sure to answer all parts. Calculate the heats of combustion for 
OneClass Consider the following reaction C2H4 g F2 g 
C2H6 C2H4 H2. If the enthalpy change for this reaction is 137 
Heats of combustion of C 2 H 4 H 2 and C 2 H 6 are 
Heat of combustion of CH4 C2H4 C2H6 are 890 1411 1550 kJ mol 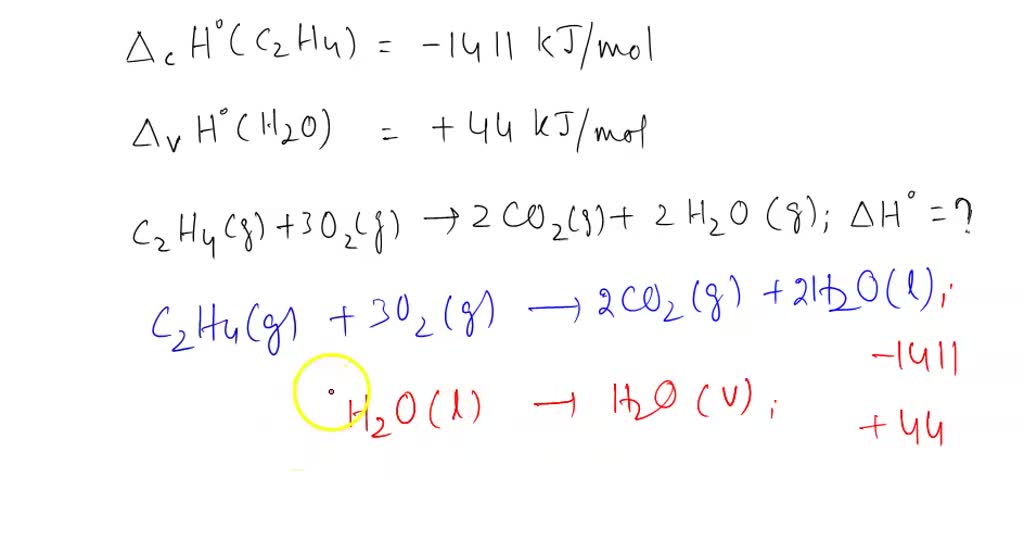
SOLVED The H combustion of ethylene C2H4 is 1411 kJ mol 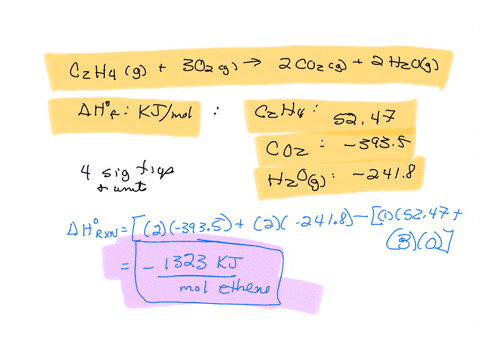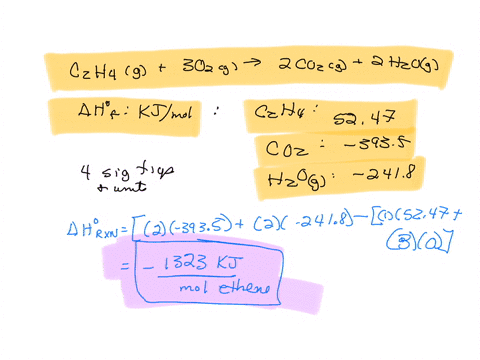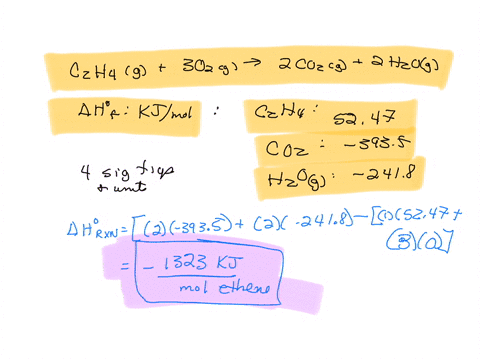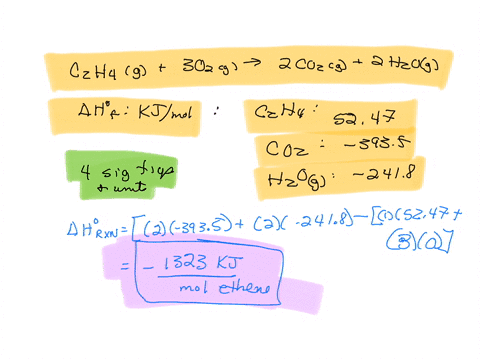
SOLVED The combustion of ethene C2H4 occurs via the reaction 
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 
The heat of combustion of ethene C2 H4 is 1409.3 kJ mol 
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol 
3.Calculate the standard enthalpy of formation of C2H4 g from the