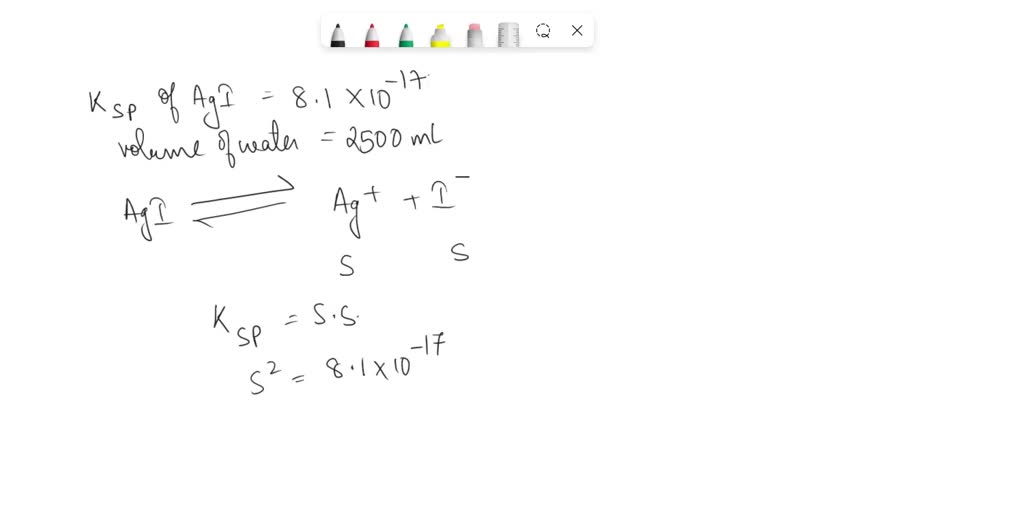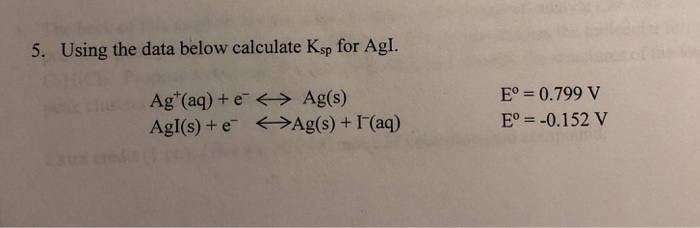Given the data 25 C Ag 1 Agl e E 0.152 V Ag Ag e
Ksp hot sale for agl
Share.
Visit »

Calculate the molar solubility of Agl in 3.0 M mathrm NH Quizlet 
Jika Ksp AgL adalah 1 5 10 16 kelarutan AgL dalam AgNO3 0 1 M 
The e.m.f. of cell Ag Agl s 0.05 M KI 0.05 M AgNO3 Ag is 
Solved The Ksp for Agl is 1 x 10 16. If you mix 300 mL of 1 
Discuss the use of ksp and solubility to seperate agcl from agbr 
Section a using the information in the preceding problem 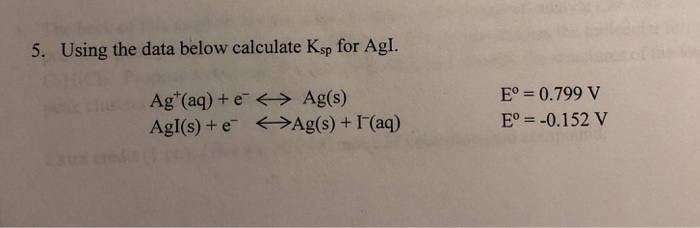
Solved 5. Using the data below calculate Ksp for Agl. Chegg 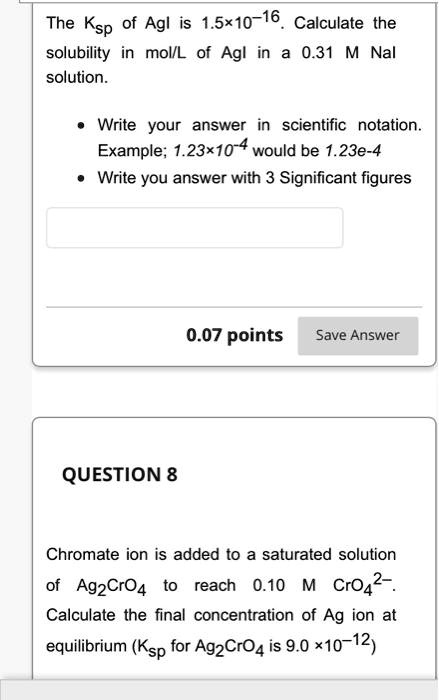
SOLVED The Ksp of AgI is 1.5x10 16. Calculate the solubility in 
SOLVED The Ksp of Agl is 1.5 x 10 16. Calculate the solubility 
The K sp of Ag 2 CrO 4 AgCl AgBr and AgI are respectively 1.1xx10 12 1.8xx10 10 5.0xx10 13 8.3xx10 17 . Which one of the following salts will precipitate last if AgNO 3 
Is silver iodide AgI soluble in water 
The solubility product of Agl at 25 C is 1.0 10 16 mol 2 L 2. The solubility of Agl in 10 
32. Ksp of AGI is 16. Standard reduction electrode potential of Ag 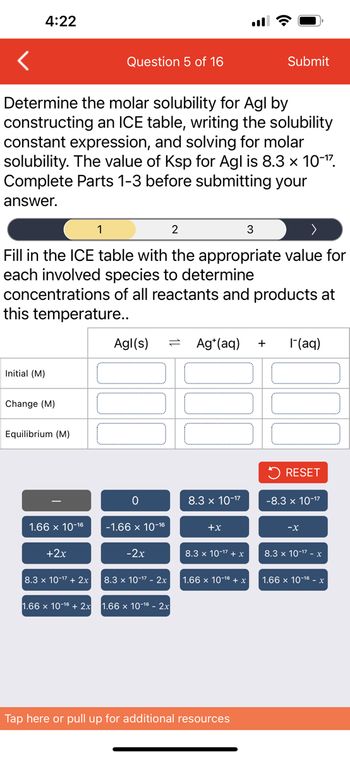
Answered Determine the molar solubility for Agl bartleby 
47. The solubility product of Agl is 
CThe solubility product of Agl at 25 C is1.0 x 10 16 mol2 L 2 
Solved Determine the molar solubility for Agl Ksp 8.3 x Chegg 
Given the data 25 C Ag Agl e E 0.152 V Ag Ag e E 
Solved Question 23 10 points The Ksp of Agl is 8.5 x 10 17 
SOLVED Calculate the molar solubility of Agl in pure water and 
Answered Determine the molar solubility for Agl bartleby 
The solubility product of Agl at 25 C is 1.0 10 16 mol2 L 2 
Use values of K sp for Agl and K f for Ag CN 2 t Quizlet 
Solved Determine the molar solubility for Agl Ksp 8.3 x Chegg 
a 1 d 4 C 3 66. The solubility product of Agl 25 C is 1 molL 
ANSWERED 5 The 22 solubility product of Agl at 25 C is 1 0 10 16 
AutoAGL on SpaceDock 
If Ksp of Agl is 8.1 10 17. The maximum amount of Agl which can 
The solubility product of AgI at 25 C is 1.0xx10 16 mol 2 L 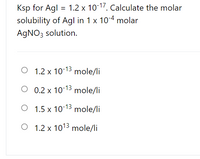
Answered Ksp for Agl 1.2 x 10 17. Calculate bartleby 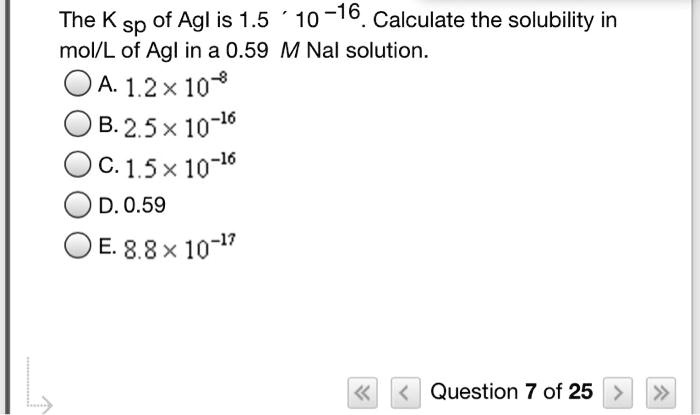
SOLVED The Ksp of Agl is 1.5 x 10 16. Calculate the solubility 
47. The solubility product of Agl is 
Solved The Ksp of Agl is 8.5 x 10 17. The Kp for the complex 
The solubility product of Agl at 25 C is 1.0 10 16 mol 2 L 2 
Ulvel the data 25 C Ag s F aq Agls e E 0.152V Ag s 
jika ksp agl 10 16 tentukan kelarutannya dalam larutan mgl2 0 
The solubility product of AgI at 25 C is 1.0xx10 16 mol 2 L 2 . The solubility of AgI in 10 4 N solution of KI at 25 C is approximately in mol L 1 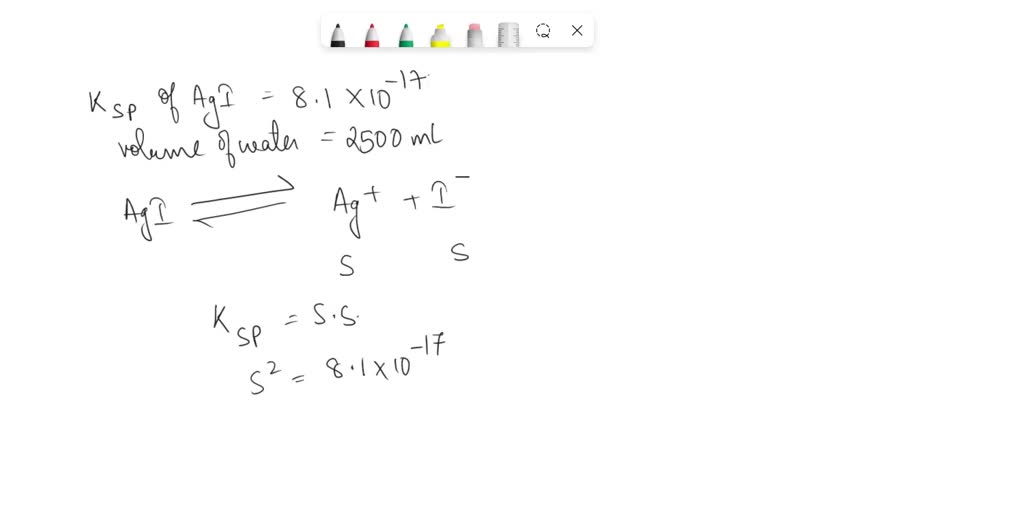
SOLVED If Ksp of Agl is 8.1 10 17 the maximum amount of Agl 
The Ksp of Ag2CrO4 AgCI AgBr and Agl are respectively 1.1x10