
Difference Between Pure and Hybrid Orbitals Compare the
Pure and hybrid hot sale orbitals in c2h4
Share. Visit »
What is the ratio of hybrid and unhybrid orbitals in the bonding
Slides22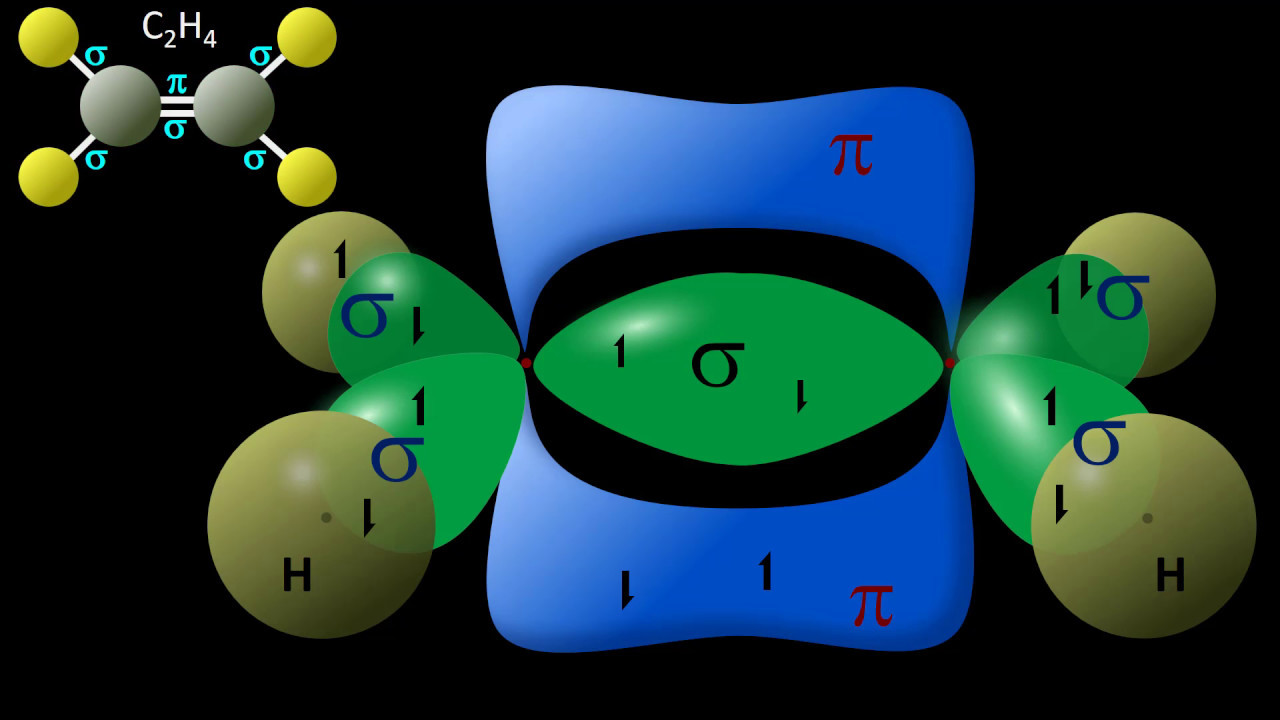
Ratio of pure and hybrid orbitals in benzene YouTube
Why do sigma bonds have hybridised orbitals and pi bonds don t
What is the hybridization and bond angle of a C2H4 molecule Quora
What is the ratio of hybrid and pure orbital in benzene Quora
Molecule Pure and hybrid orbitals invou List B C2H6 hitals
Why do sigma bonds have hybridised orbitals and pi bonds don t
2.2. Hybrid orbitals Organic Chemistry 1 An open textbook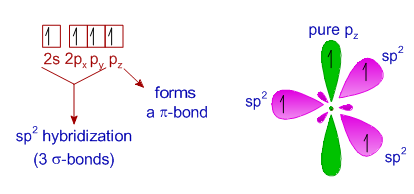
Hybridization Examples in Chemistry Types sp sp2 sp3 sp3d sp3d2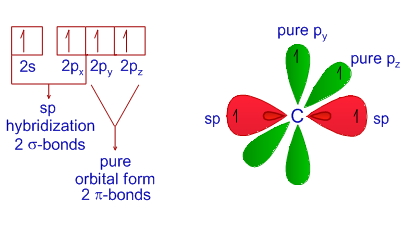
Hybridization Examples in Chemistry Types sp sp2 sp3 sp3d sp3d2
Ratio of pure and hybrid orbitals in benzene
Slides22e
Why didn t the 3rd P orbital of carbon in C2H4 take part in
What are those smaller lobes in hybrid orbitals Quora
The ratio of pure and hybrid orbitals H 2 C CH CH CH 2
find the ratio of no.of. pure and hybrid orbitals in benzene with
The ratio of pure orbitals to hybridized orbitals in ethylene is a
The ratio of pure orbitals to hybridized orbitals in ethylene is
Why didn t the 3rd P orbital of carbon in C2H4 take part in
Why do the two empty orbitals 2p in ethene overlap sideways and
Trick to Find Ratio of Hybrid and Pure Orbitals
Which hybridized orbital has the maximum electron density sp3 sp2
Telugu The ratio of pure orbitals to hybridized orbitals in ethylene
Trick to Find Ratio of Hybrid and Pure Orbitals YouTube
Orbital hybridisation Wikipedia
Slides22e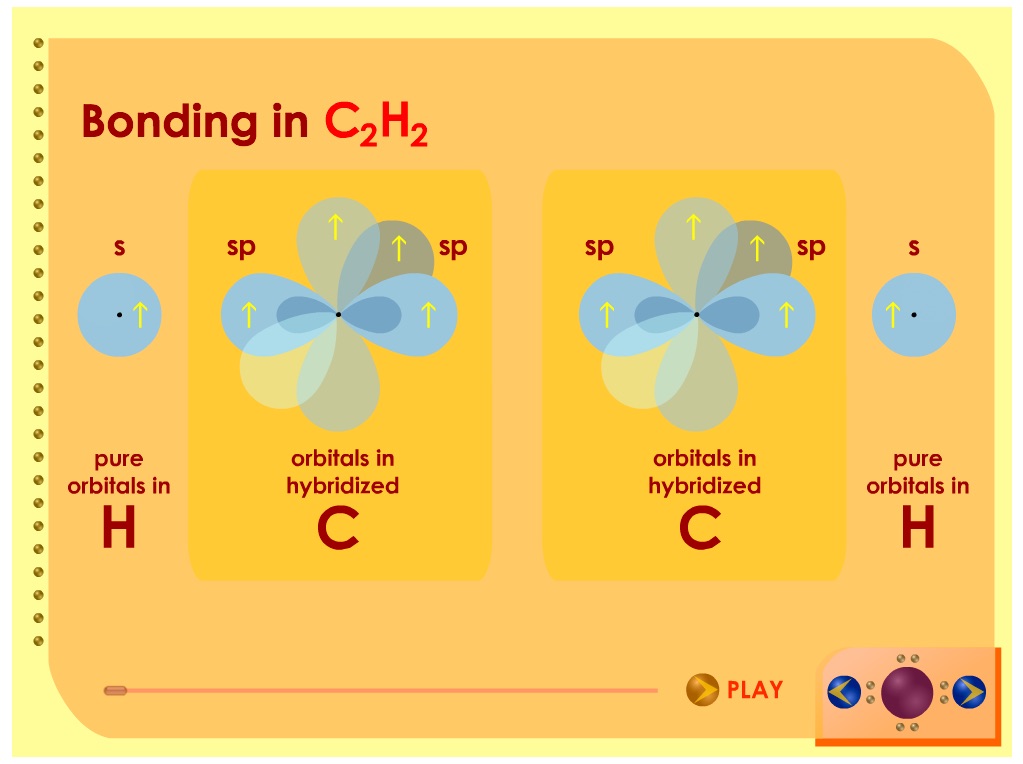
Hybrid Orbitals Interactive Learning Center Diliman
Hybrid orbitals forming molecular orbitals ECHEMI
Hybridization NEB Grade 11 Notes Physical Chemistry Sajha Notes
sp hybridization video Khan Academy
Slides22
How to determine hybridization in C2H6 Quora
Slides22e
Molecule Pure and hybrid orbitals invou List B C2H6 hitals
Molecule Pure and hybrid orbitals invou List B C2H6 hitals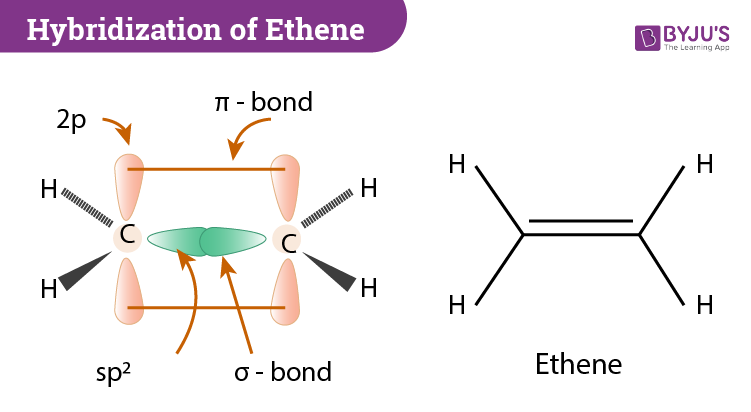
Hybridization of C2H4 Ethene Hybridization of Carbon in C2H4
2.2. Hybrid orbitals Organic Chemistry 1 An open textbook
Why do sigma bonds have hybridised orbitals and pi bonds don t