Heat of combustion of CH 4 C 2 H 4 C 2 H 6 are 890 1411 and 1560
Heat of combustion of hot sale ch4 c2h4 and c2h6 are
Share. Visit »
Heat of combustion of C 2H 4 is 337 K Cal. If 5.6 lit O 2 is used
ntif enthalpies of formation for c2h4 g co2 g and h2o l at
Heat of combustion of CH4 C2 H6 C2 H4 and C2 H2 gases are
Answered Using the following equation and the bartleby
Which of the following is best fuel Given heats of combustion of CH4
Heat of combustion of CH 4 C 2H 4 C 2H 6 are 890 1411 and 1560 kJ mol respectively. Which has the lowest calorific fuel value in kJ g
Heat of combustion of CH4 C2H6 and C3H8 are respectively
Heat of combustion of CH4 C H and C. He are 890 1411 and 1560
Enthalpies of combustion of CH 4 C 2H 4 and C 2H 6 are 890
heat of formation of ethane 20.2kcal mol and heat of atomisation
Heat of combustion of mathrm CH 4 mathrm C 2 mathrm H 4
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g
enthalpies of combustion of CH4 and C2H6 are 890 and 1559 kilo
What is the standard enthalpy change at 298K for the reaction CH4
Heats of combustion of C 2 H 4 H 2 and C 2 H 6 are
Be sure to answer all parts. Calculate the heats of combustion for
Solved 5. Determine the enthalpy of combustion of methane Chegg
Heat of combustion of CH 4 C 2 H 6 C 2 H 4 and C 2 H 2 gases are 212.8
Given that standard heat enthalpy of mathrm CH 4 mathrm C 2 mathrm H 4 and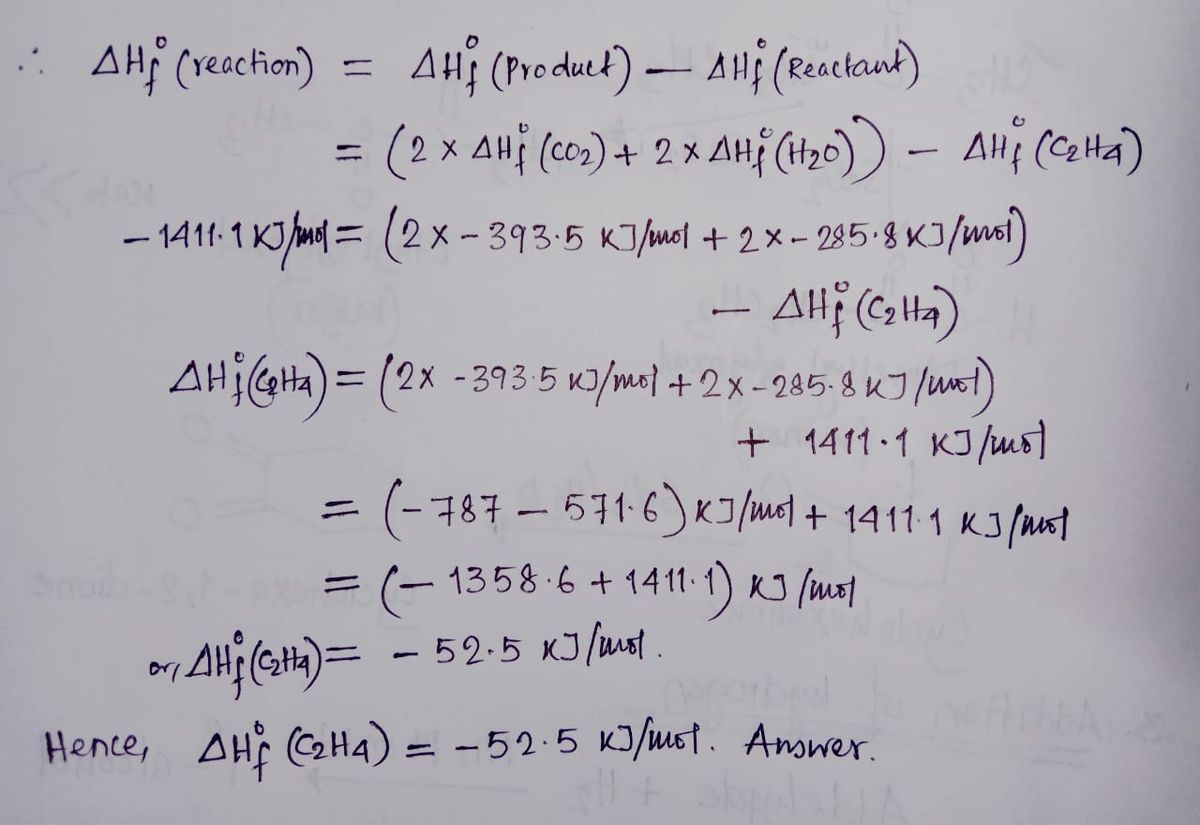
Answered The standard enthalpy of combustion of bartleby
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol
Solved 1. A gaseous fuel containing methane CH4 and Chegg
Uncal 39. The heat of combustion of ethene C H. is 1409.3 kJ
3. The standard enthalpies of formation of CH4 CO2 and H20 are
EWD 111 WE LLAVA 50. Heat of combustion of CH4 C2H4 C2H6 are
EWD 111 WE LLAVA 50. Heat of combustion of CH4 C2H4 C2H6 are
Heat of combustion of CH4 C2H4 C2H6 are 890 1411 1550 kJ mol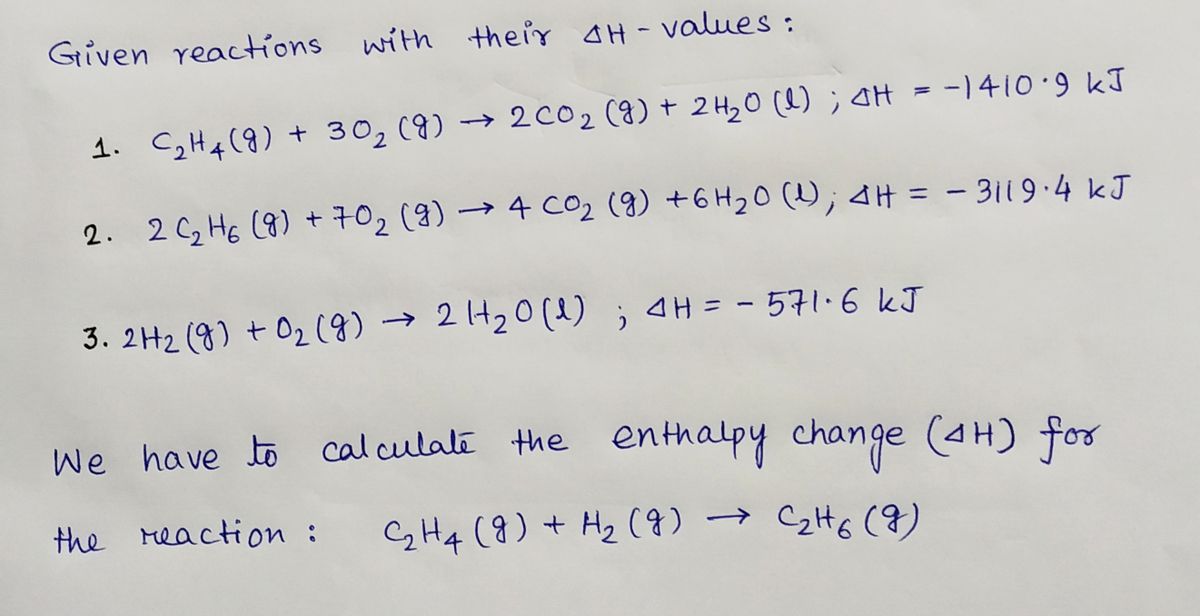
Answered Calculate the enthalpy change for the bartleby
The standard molar heat for formation ofethane carbondioxide and
Odia Heat of combustion of CH4 C2H4 C2H6 are 890 1411 and 1560
Concentration profiles of C2H6 C2H4 and C2H2 in H2 CH4 C3H8 O2 Ar
Heat of combustion of CH4 CH C H and CH gases are 212.8
EWD 111 WE LLAVA 50. Heat of combustion of CH4 C2H4 C2H6 are
CH4 C2H2 C2H6 is entalpy of combustion 890 1411 1560 kj mol so
Be sure to answer all parts. Calculate the heats of combustion for
Heat of combustion of CH4 C2H6 and C3H6 are respectively 210 368
The relationship of CH4 C2H4 and C2H6 volume fraction with coal
CH4 C2H6 and C3H8 consumption of each reaction involved mixture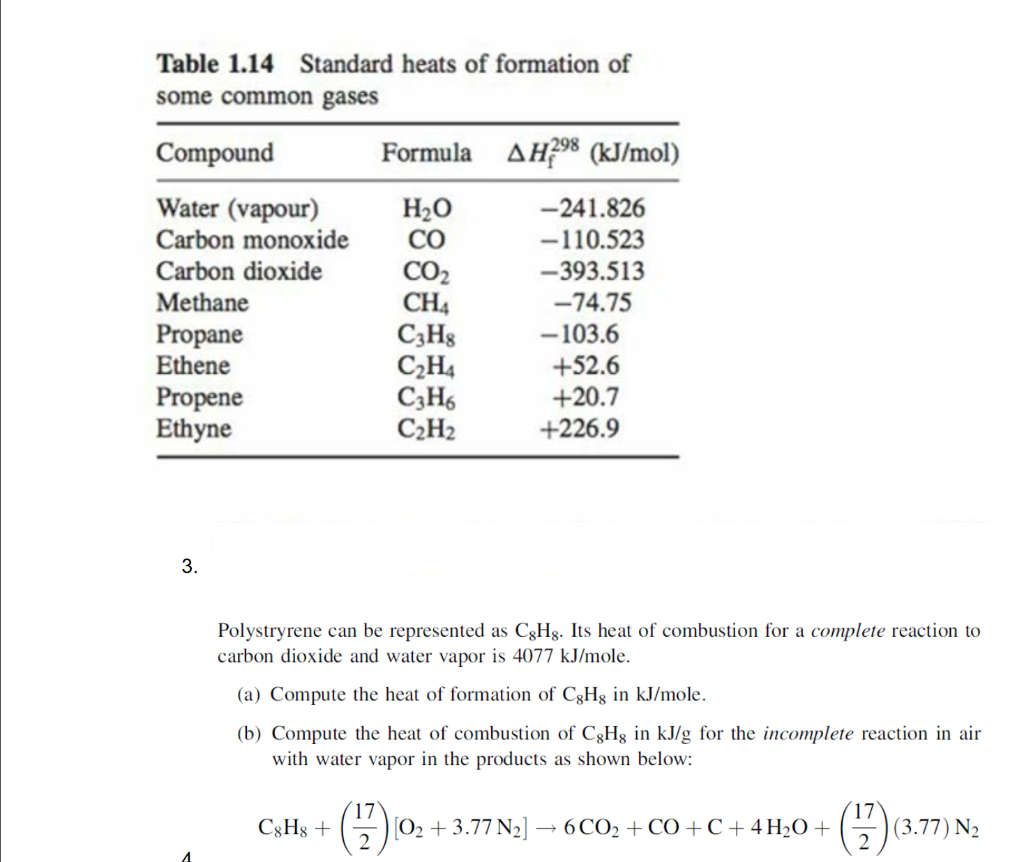
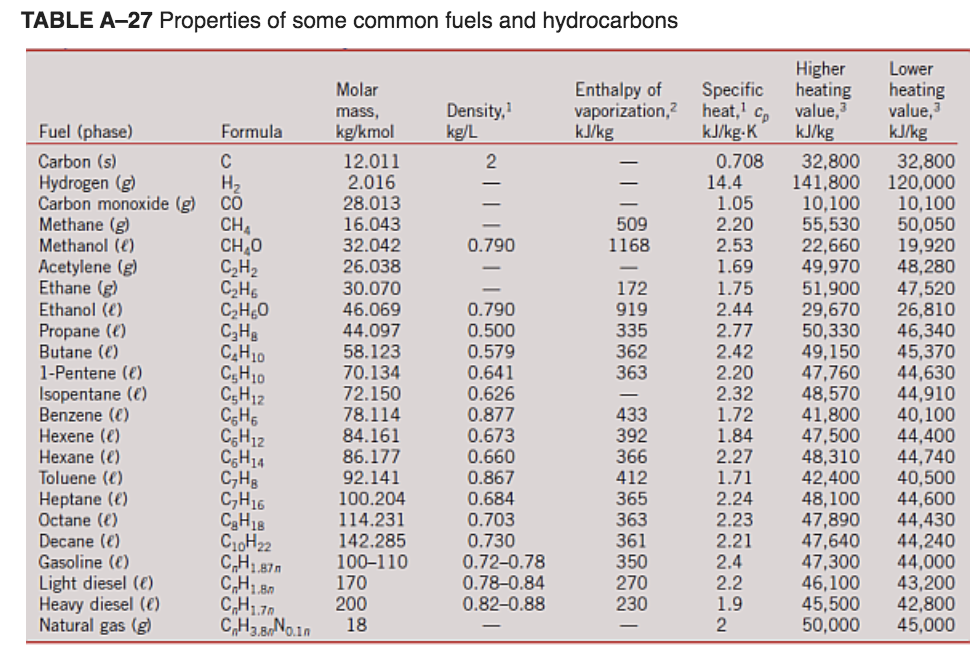
Solved Table 1.13 Heats of combustion of selected fuels at Chegg