Lewis Structure for C2H4
How many bonds hot sale are in c2h4
Share.
Visit »
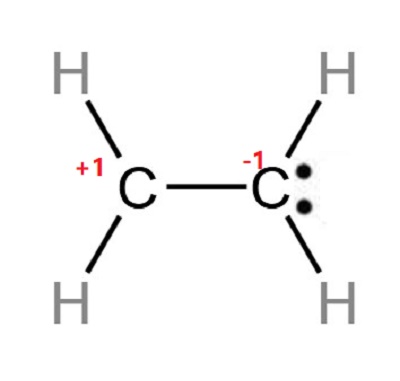
What is the Lewis structure of Ethylene Chemicalbook 
ethene molecule C2H4 Lewis dot cross electronic diagram sigma pi 
Draw the Lewis structure for C2H4 whose skeletal structure is 
How many sigma bonds are formed in ethene Quora 
Draw and explain the Lewis structure of C2H4. Homework.Study 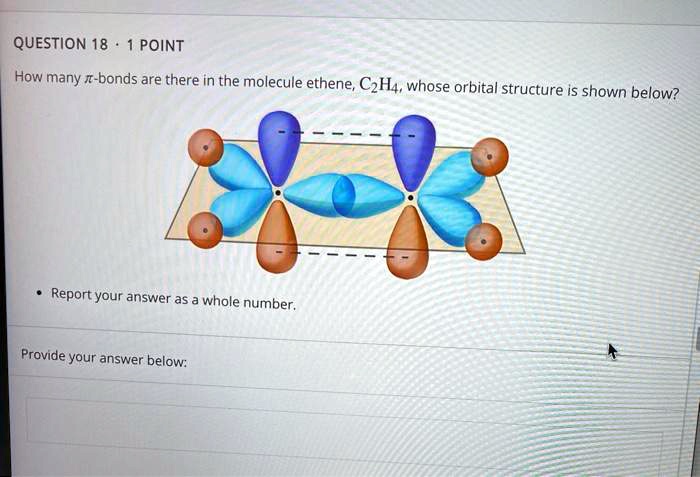
SOLVED QUESTION 18 POINT How many bonds are there in the 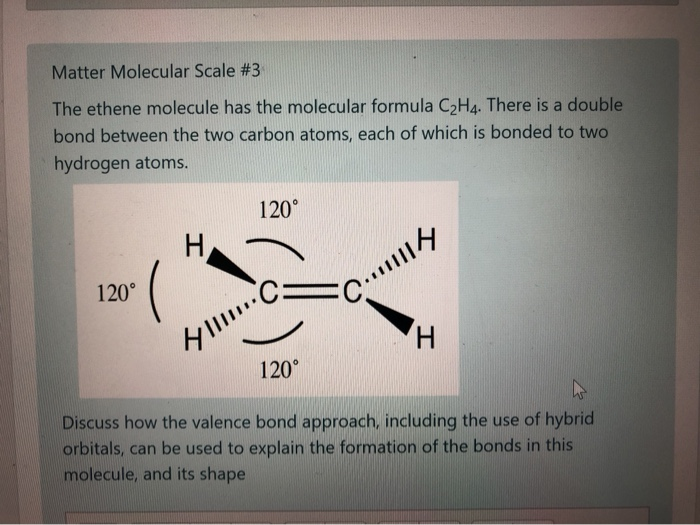
Solved Matter Molecular Scale 3 The ethene molecule has the 
SOLVED How many sigma and pi bonds are present in C2H4 a.5 pi 
C2H4 Hybridization Ethylene Facebook 
Draw the Lewis dot structure for C2H4 and a second structure 
How many sigma bonds and pi bonds are present in C2H4 Ethene 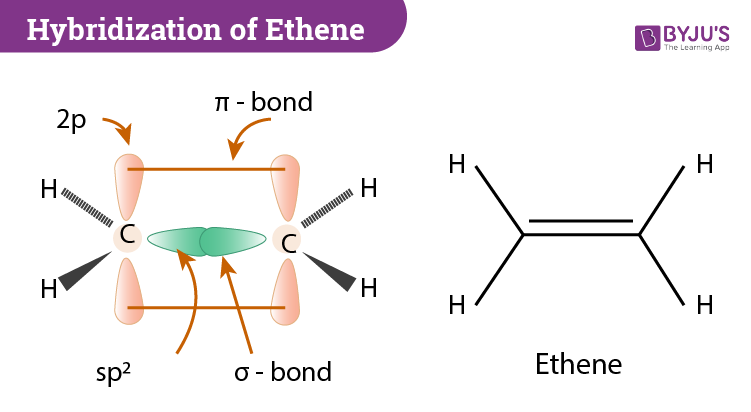
Hybridization of C2H4 Ethene Hybridization of Carbon in C2H4 
Solved Describe the bonding in ethylene C2H4 in terms of 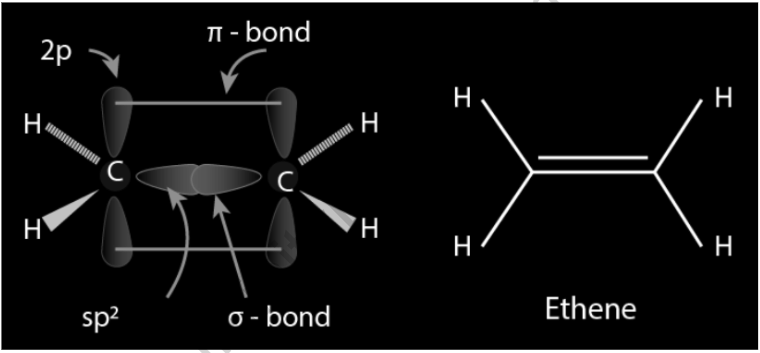
Structure of Double Bond Ethene Infinity Learn by Sri Chaitanya 
How many sigma bond and pi bond are there in C2H4 Explain with 
Draw Lewis structures for the following molecular formulas C2H4 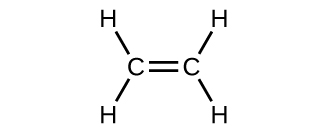
8.3 Multiple Bonds Chemistry 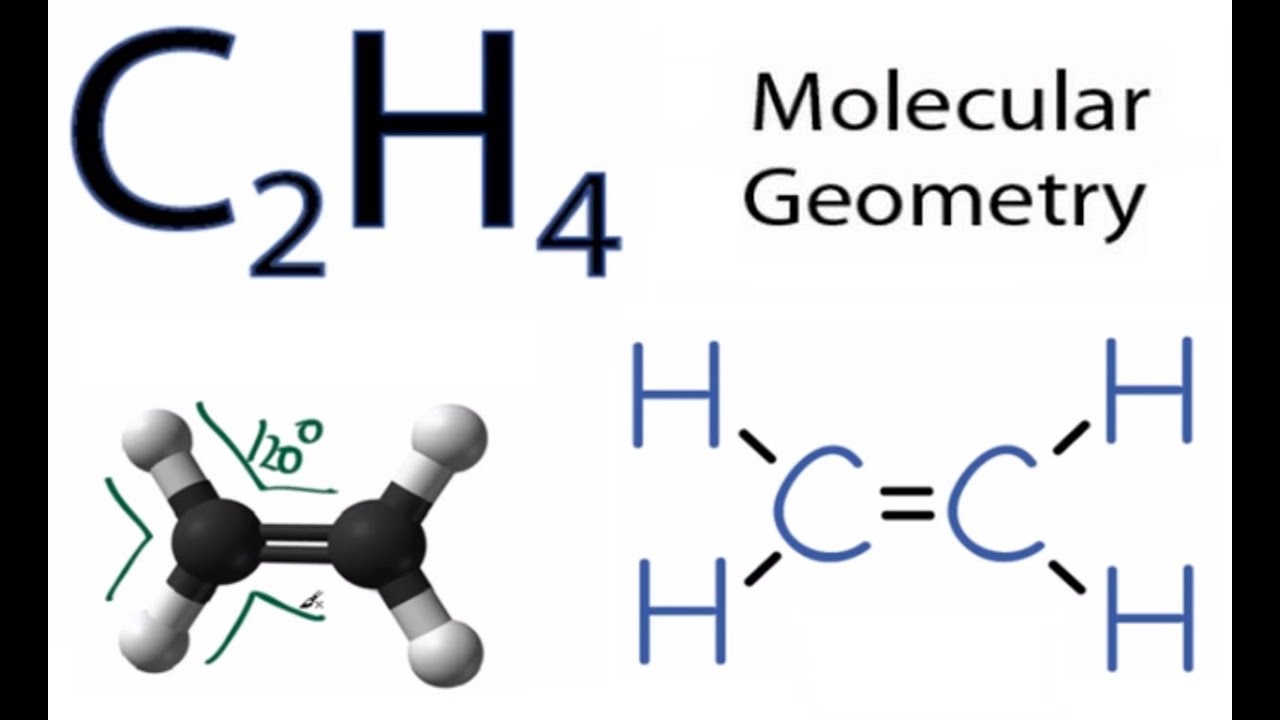
C2H4 Molecular Geometry Shape and Bond Angles 
Draw Lewis structures for the following molecular formulas C2H4 
Orbital Overlap Diagram for C2H4 Ethene acetylene double bond 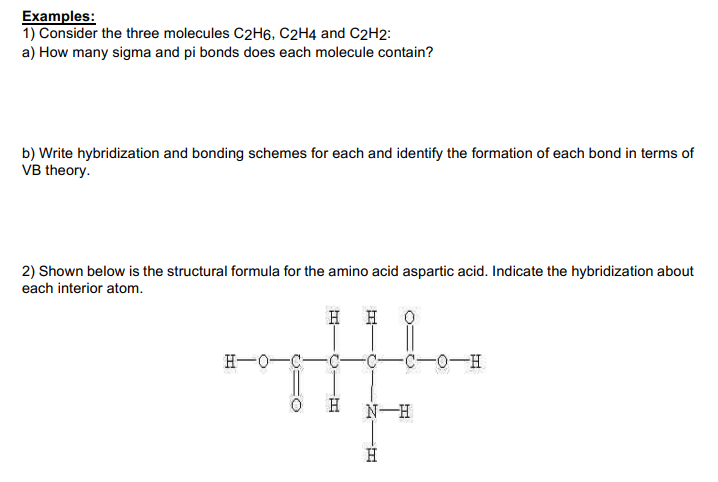
Solved Examples 1 Consider the three molecules C2H6 C2H4 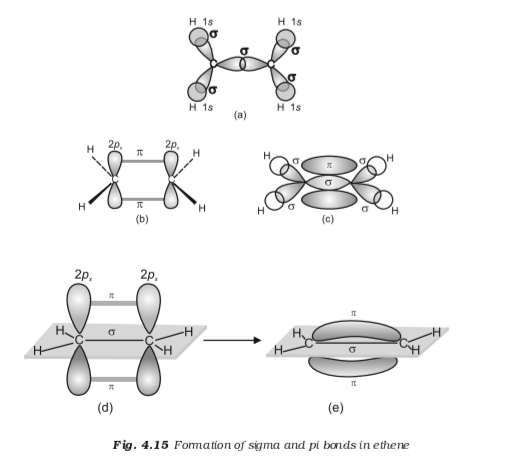
C2H4 Hybridization 
Explain the geometries of C2H2 C2H4 C2H6 based on VBT 
What are the total number of sigma and pi bonds in the following 
13.2. Molecular orbitals for ethene Organic Chemistry II 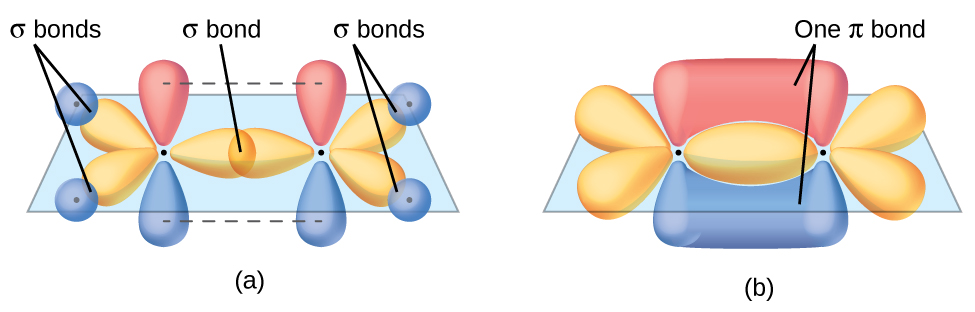
8.3 Multiple Bonds Chemistry 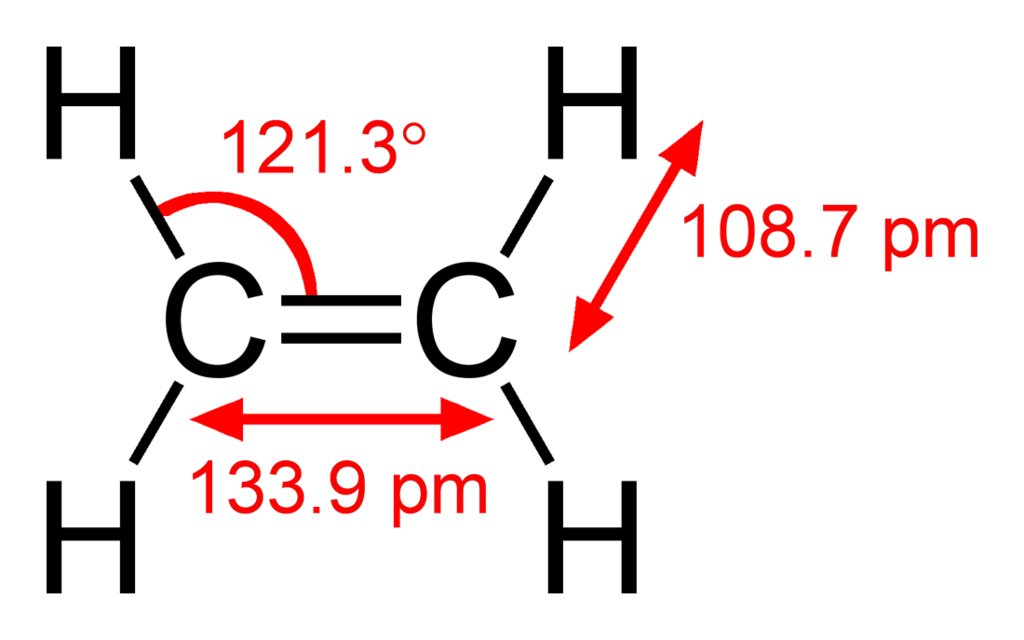
C2H4 Molecular Geometry and Bond Angles 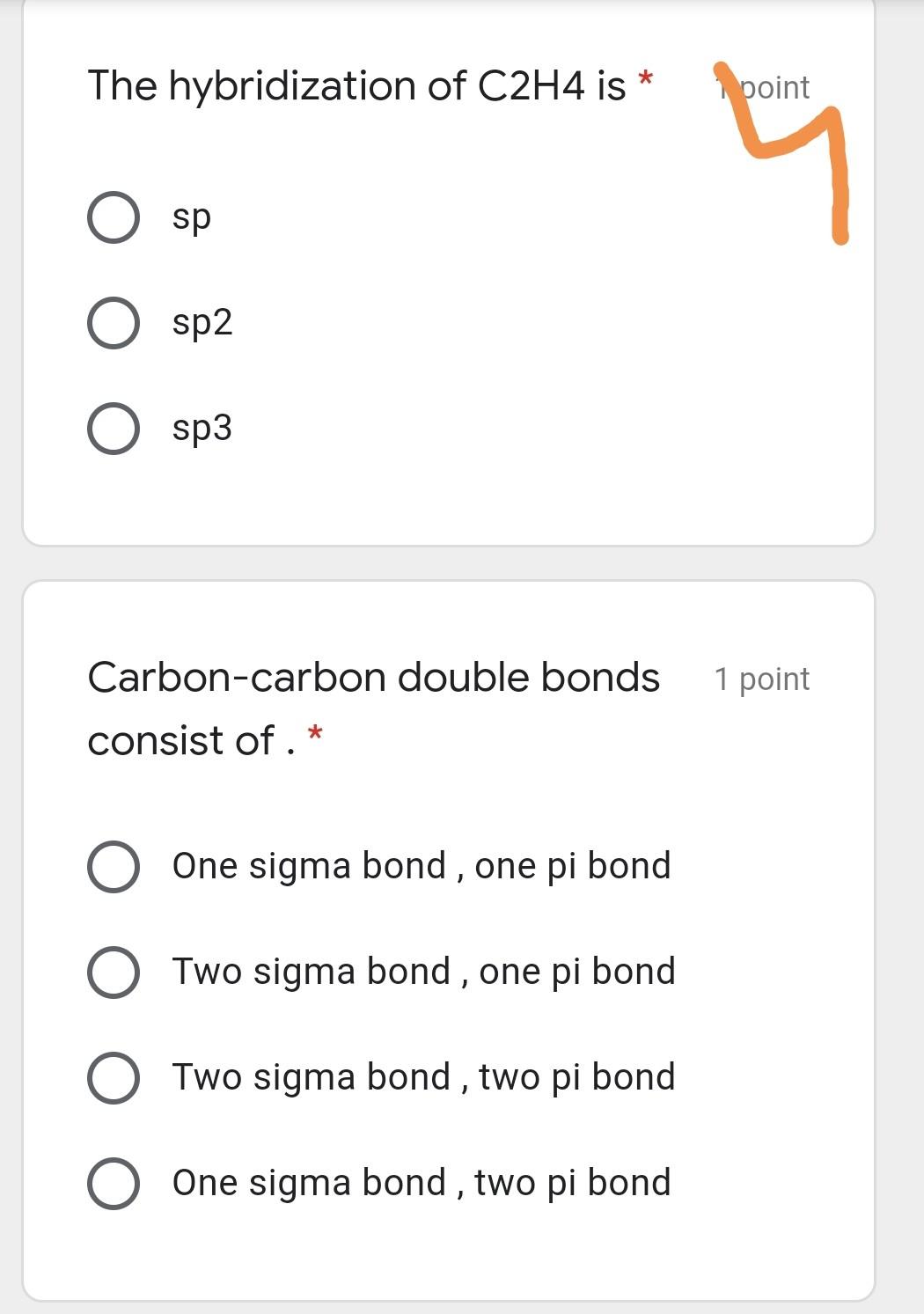
Solved The hybridization of C2H4 is 1 point sp O sp2 O sp3 
indicate the number of pi bonds in C2H4. Homework.Study 
Ethene with the molecular formula C 2 H 4 hasA. 5 covalent 
How many sigma bonds and pi bonds are present in C2H4 Ethene 
Covalent bonding in an ethene molecule C2H4 
How many sigma and pi bonds are present in ethene Quora 
Draw diagrams showing the formation of a double bond and a triple 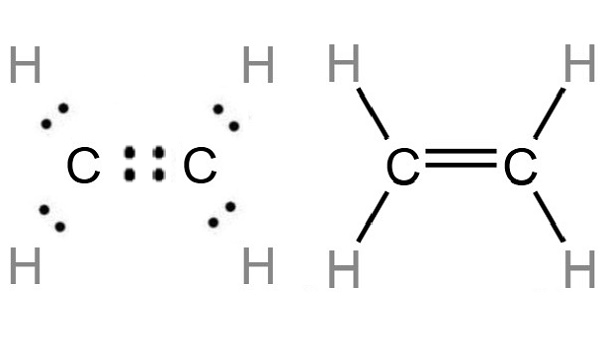
What is the Lewis structure of Ethylene Chemicalbook 
Answered Consider the ethylene molecule C2H4. bartleby 
What is the total number of sigma and pi bonds in C2H4 Ethene 
a CH4 covalent bond and bond hybridized orbital b C2H6 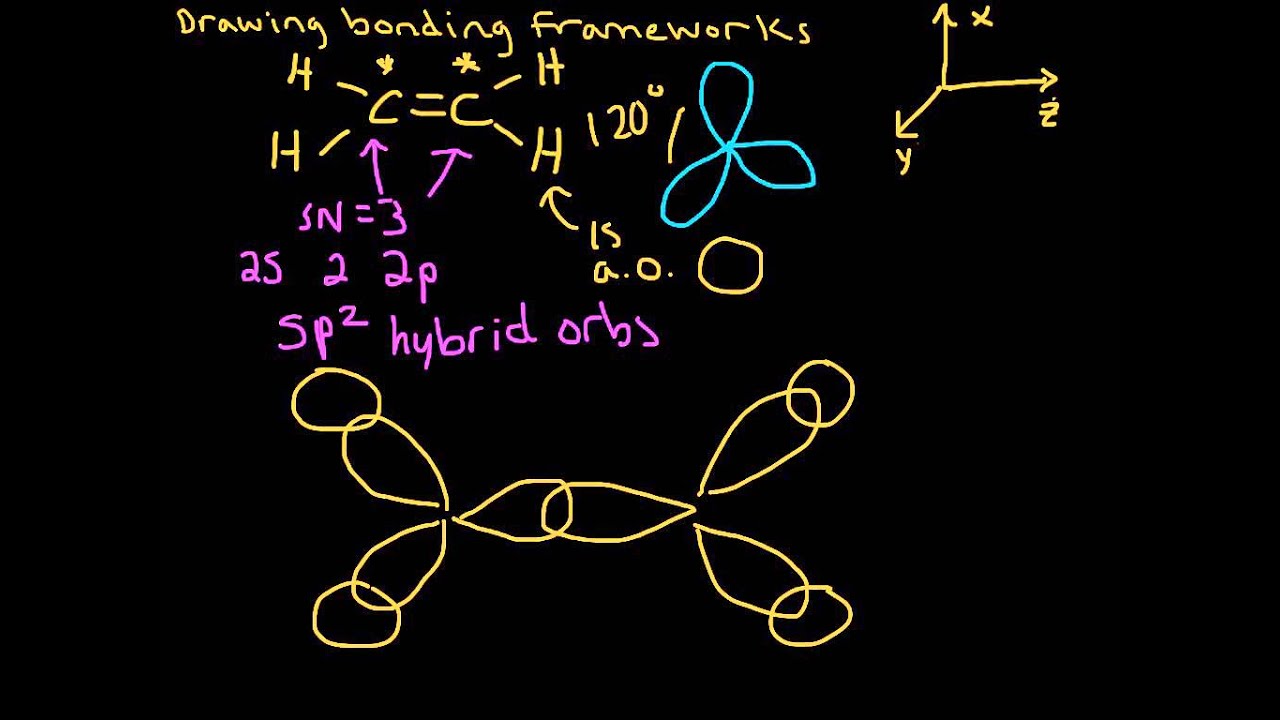
Sigma and Pi Bonding in Ethene C2H4 Hybridization

































