Solved Use the bond energies provided to estimate AH rxn for
Bond hot sale energy c2h4
Share. Visit »
Solved Use Lewis structures and bond energies to calculate Chegg
Calculated energy profile for the dissociation of one C H bond of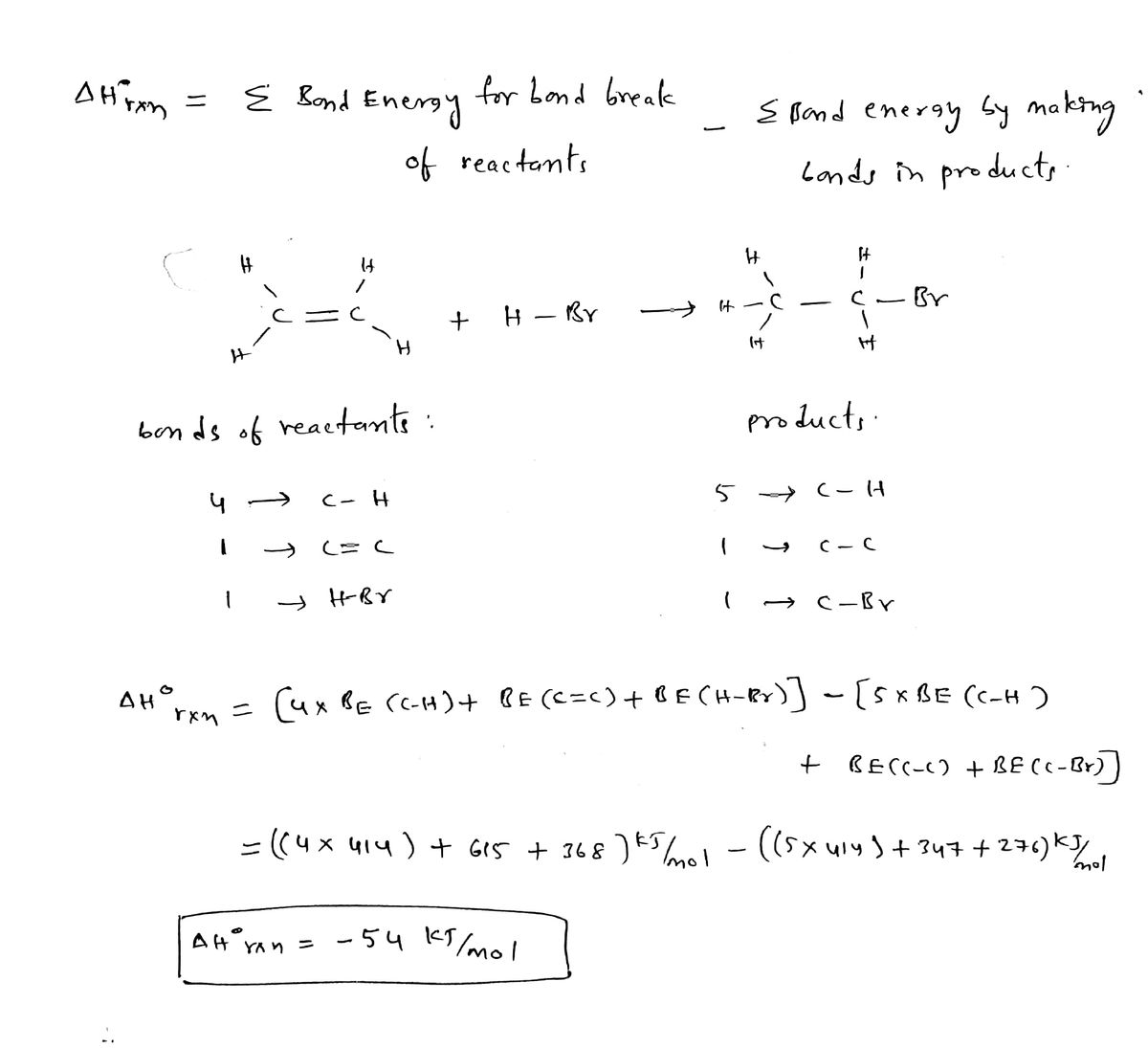
Answered Use the molar bond enthalpy data in the bartleby
OneClass Consider the following reaction C2H4 g F2 g
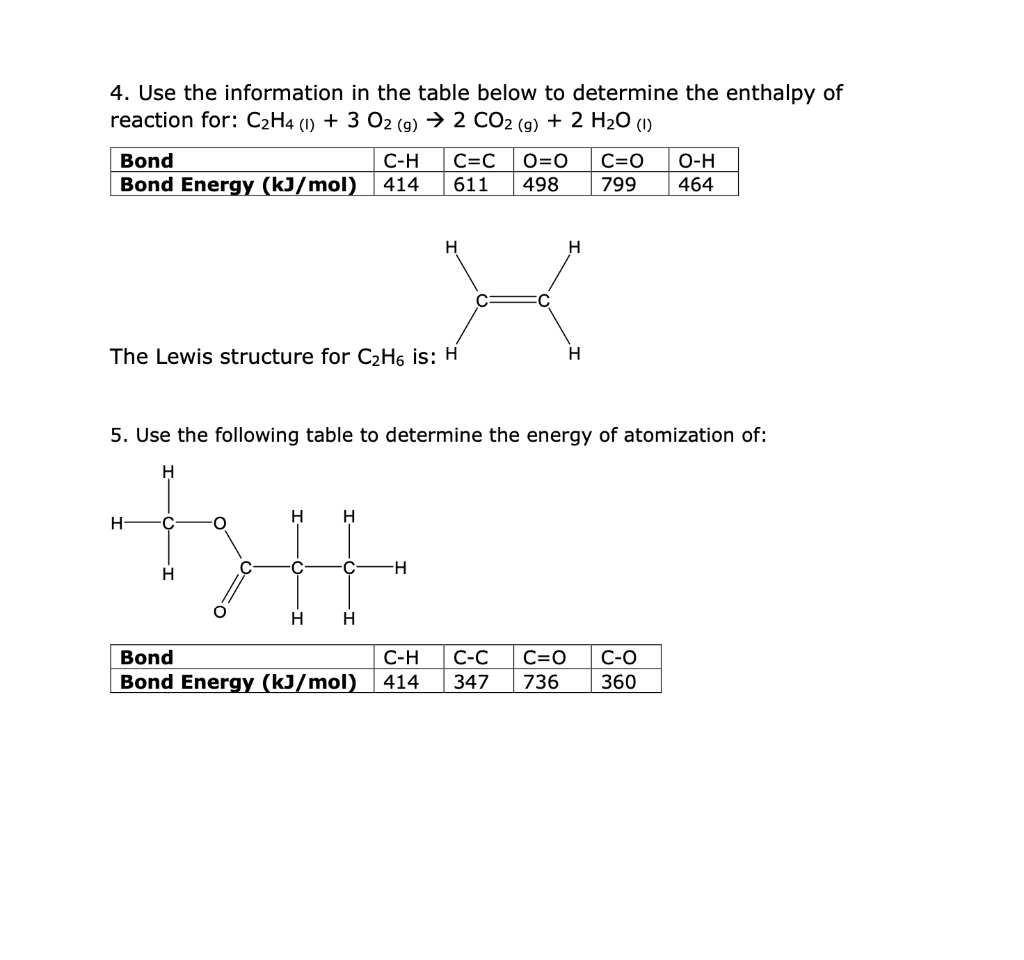
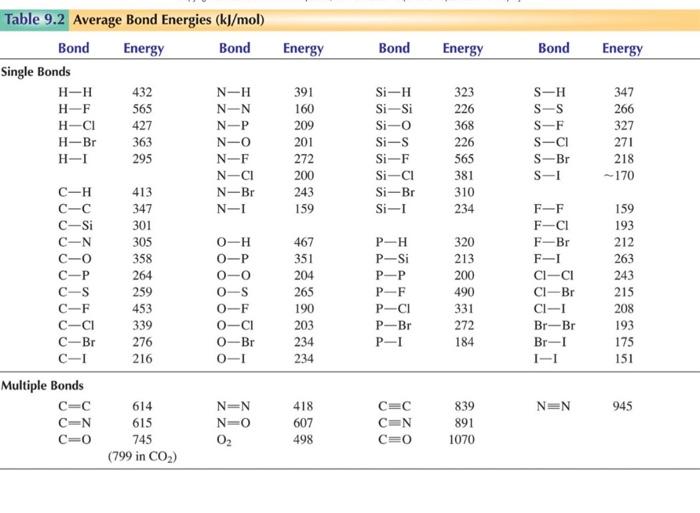
Solved 4. Use the information in the table below to Chegg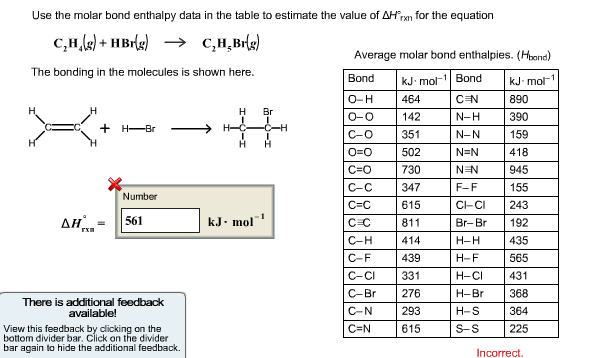
Solved use the molar bond enthalpy data in the table to Chegg
Solved Use Lewis structures and bond energies to calculate Chegg
Solved Using the values of bond energy from the table above H2(g) → C2H6(g).jpg)
1.2.6 Bond Enthalpies. ppt download
IB SL Chemistry Mrs. Page ppt download
Chapter 9 Reaction Energetics
The Bond energy of C C C C C H and H H are 147 83 99 and 104 kcal mol respectively then calculat
Delta H 1 C 2 H 4 12.5 kcal Heat of atomisation of C 171 Kcal Bond energy of H 2 104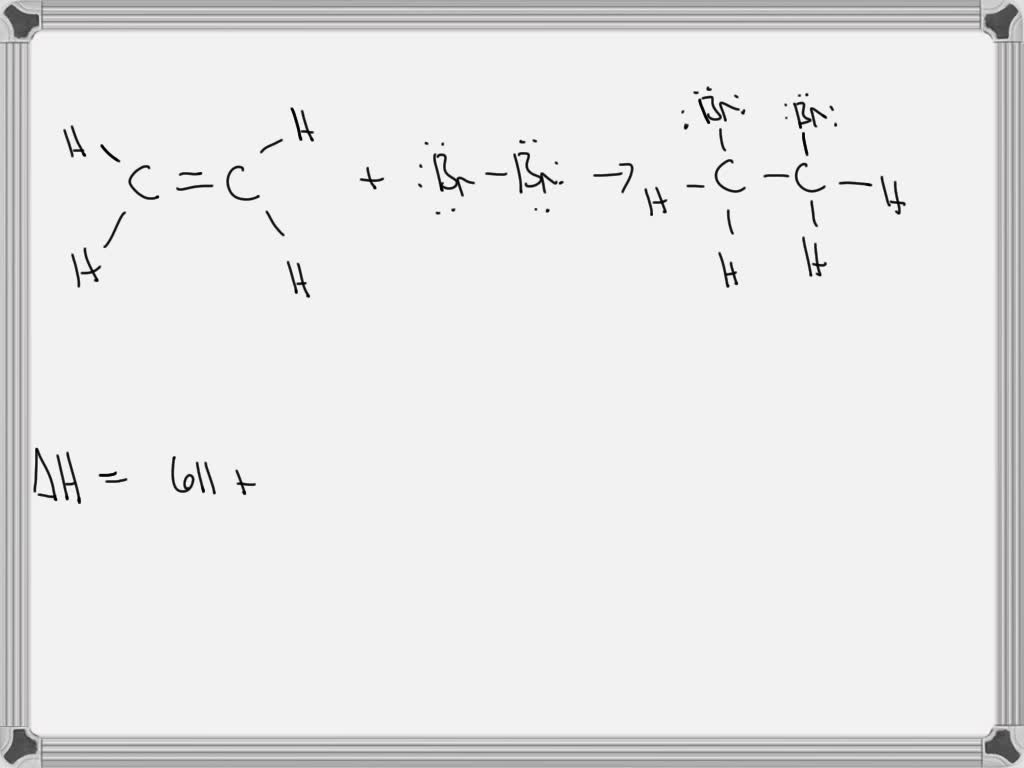
SOLVED Consider the bromination of ethene C2H4 g Br2 g
Hf C2H4 12.5 kCal Heat of atomisation of C 171 kCal Bond
Enthalpies of Reactions Using Average Bond Enthalpies Chemistry Tutorial
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol
Use bond energies to confirm that the complete combustion of
Using the required bond energies data calculate the heat of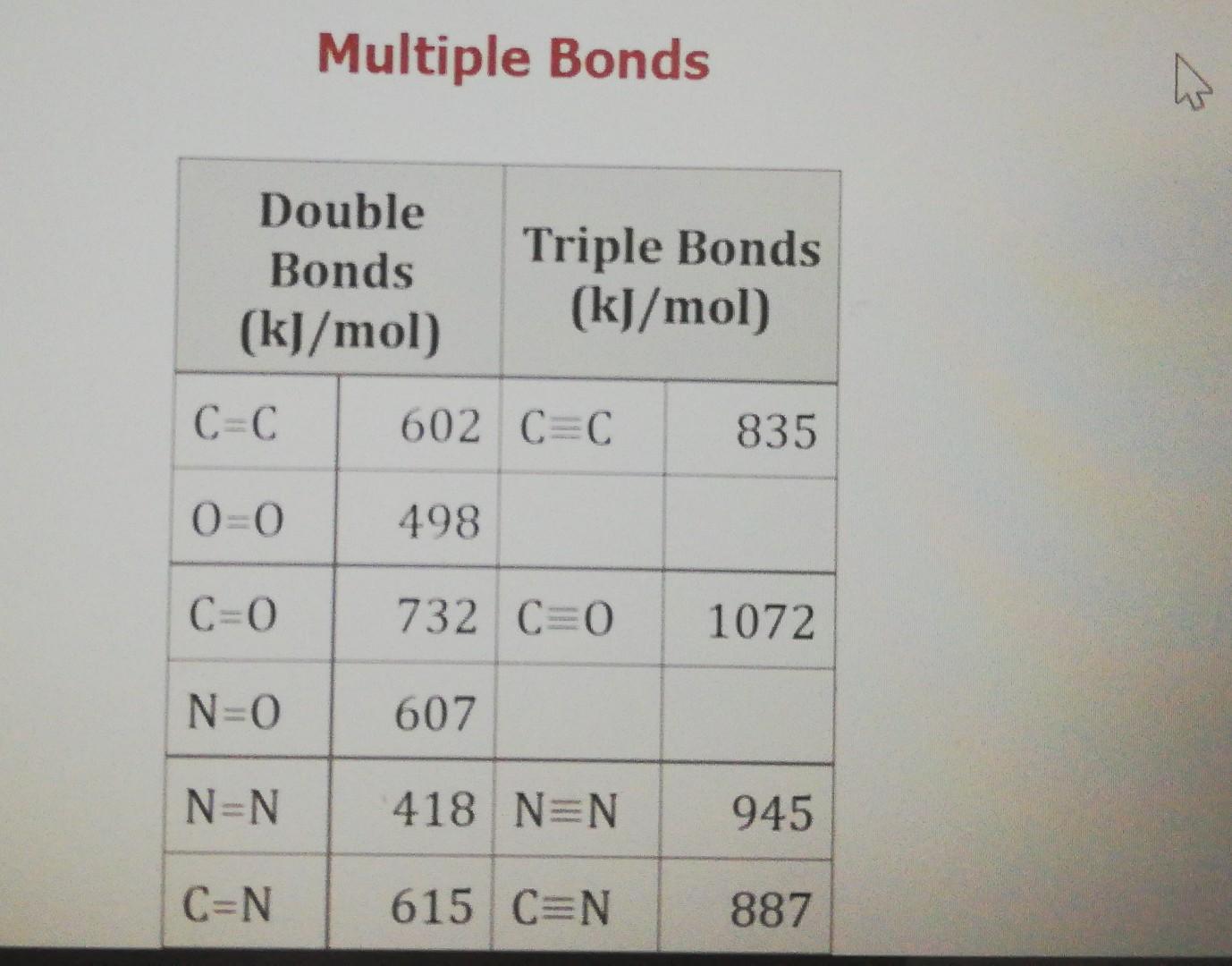
Solved Using the values of bond energy from the table below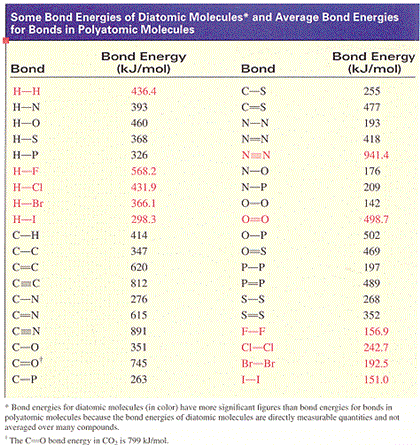
Solved H2 g C2H4 g C2H6 g a Estimate the enthalpy of Chegg
24. Delta Hf C2H4 12.5 kcal Heat of atomization of c 171 kcal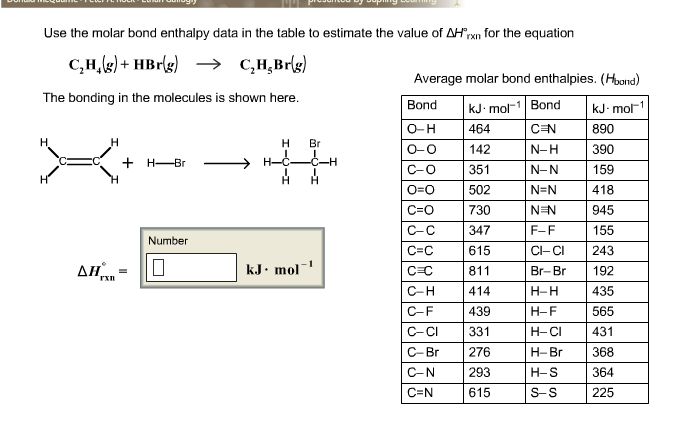
Solved Use the molar bond enthalpy data in the table to Chegg
18. Calculate the enthalpy of hydrogenation of C2H2 g to C2H4 g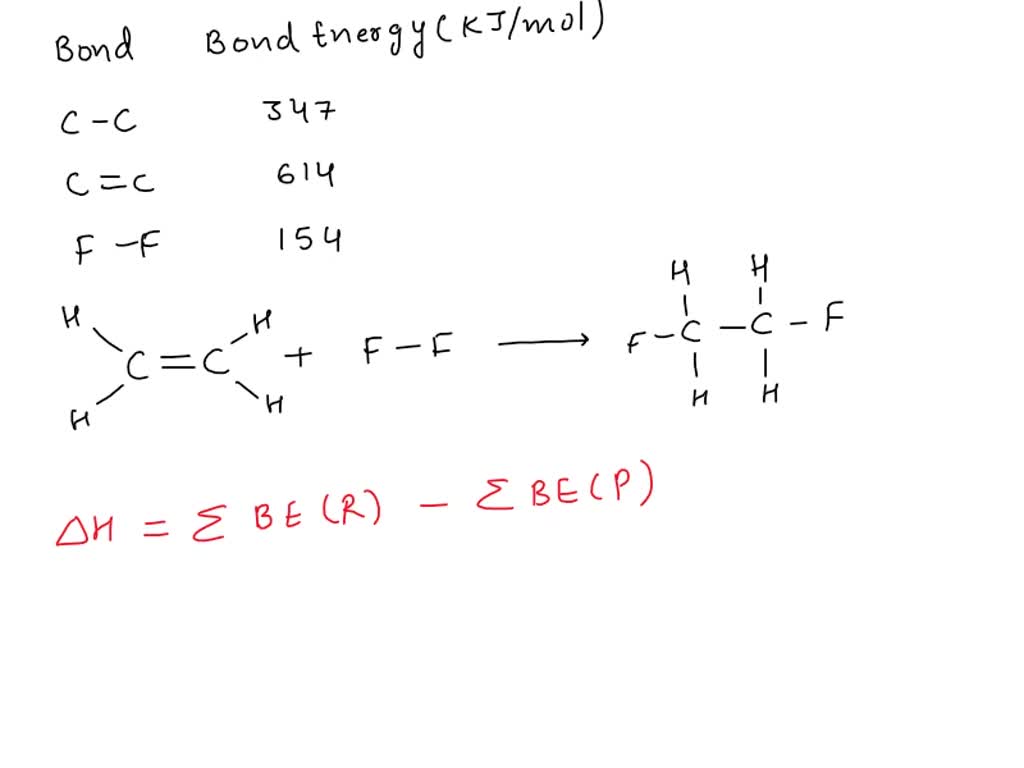
SOLVED Consider the following reaction C2H4 g F2 g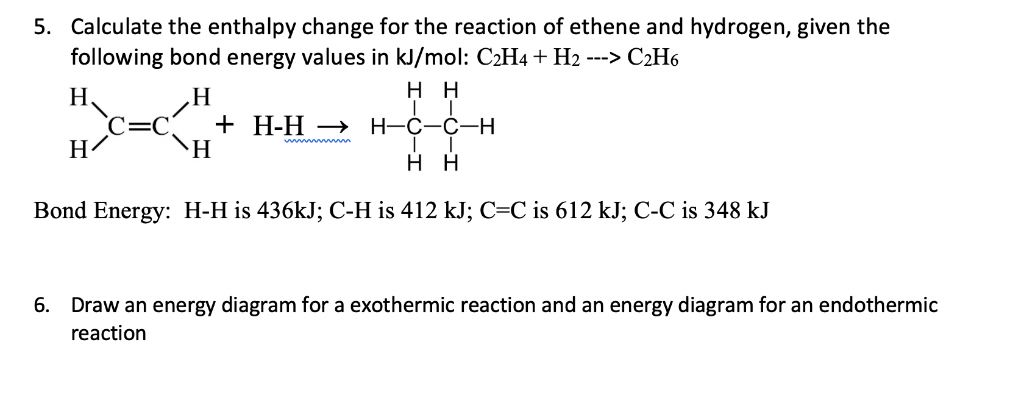
Solved Calculate the enthalpy change for the reaction of Chegg
Solved Use bond energy values to estimate Hfor the following
Using the table of average bond energies determine the total bond
Answered Acetylene C H2 can be converted to bartleby
Solved 22. Use average bond energies from the table below to
Calculating the Carbon Iodine Bond Energy in Iodination of Ethene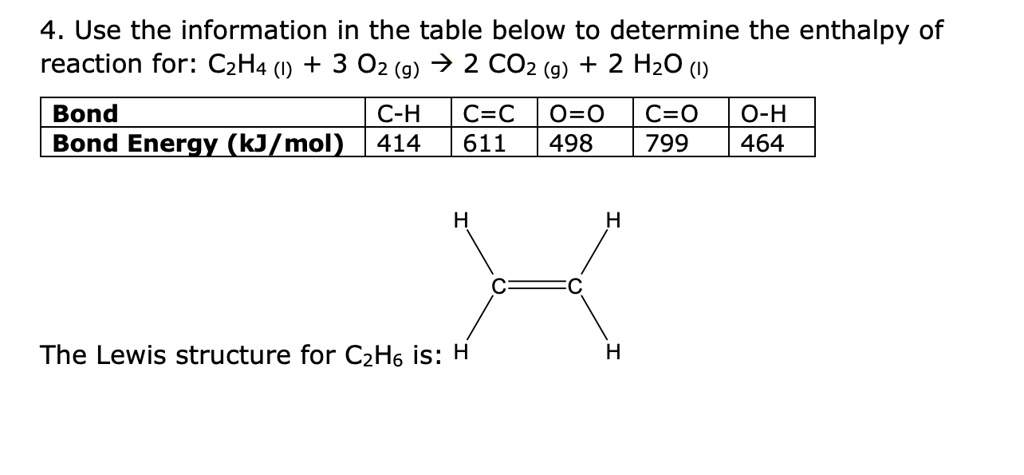
SOLVED Use the information in the table below to determine the
Solved Question H2 g C2H4 g C2H6 8 BE C H 413
Answered For the combustion reaction of ethylene bartleby
OneClass Consider the following reaction C2H4 g F2 g
From the following bond energies H H bond energy 431.37kJ mol
Solved Estimate the enthalpy change for the reaction C2H4 g
Determination of the Rh C bond energy for C2H2 and C2H4 reactive
The enthalpy change for the following reaction is 137 kJ. Using