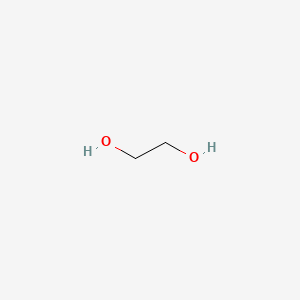
How many grams of ethylene glycol C2H4 OH 2 must be added to
C2h4 oh 2 vapor hot sale pressure at 60 degrees celsius
Share. Visit »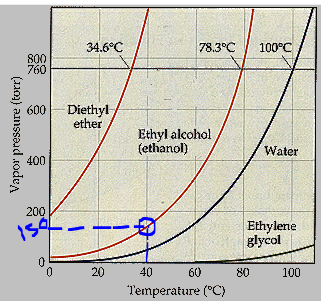
Answered 34.6 C 78.3 C 100 C 800 760 600 bartleby
Answered Explaining Vapor Pressure in Terms of bartleby
Ethylene glycol 107 21 1
Formerly ethylene glycol is mixed with the water in a car radiator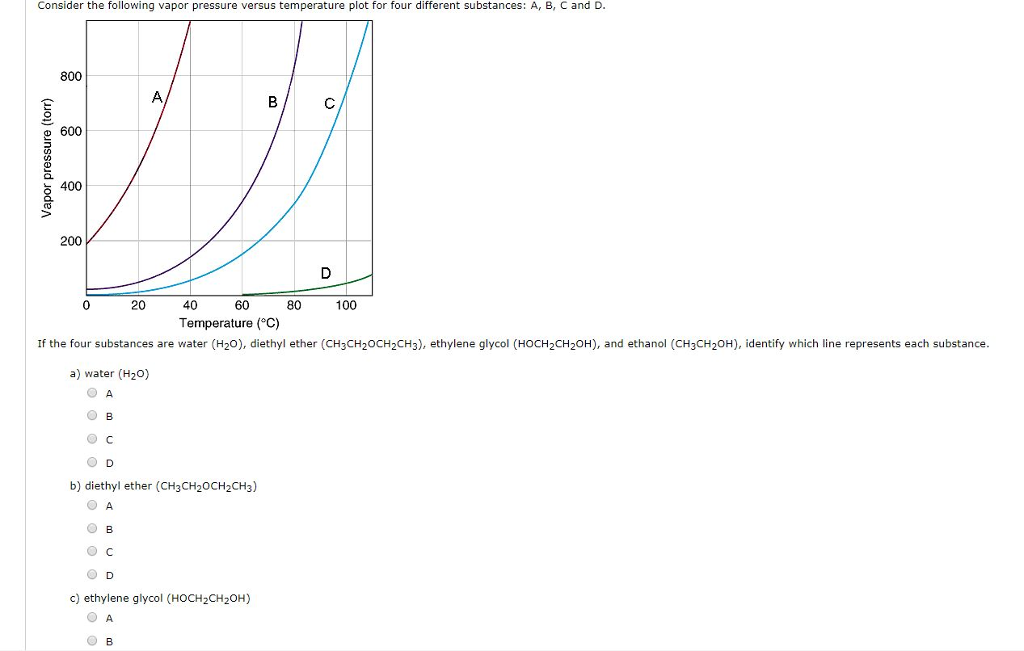
Solved Consider the following vapor pressure versus Chegg
What is the vapor pressure in mm Hg of the following solu tions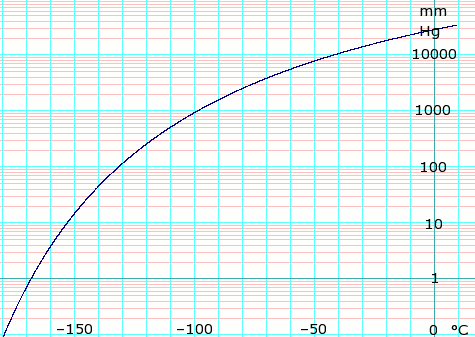
Ethylene data page Wikipedia
Pressure p liquid mole fraction x C2H4 and vapor mole fraction y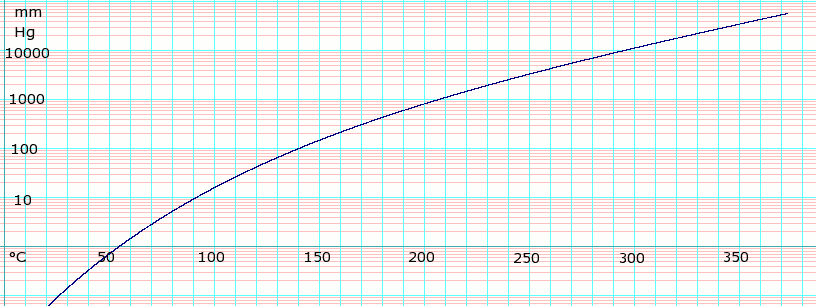
Ethylene glycol data page Wikipedia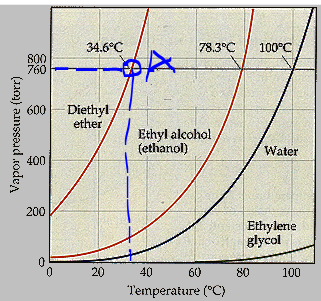
Answered 34.6 C 78.3 C 100 C 800 760 600 bartleby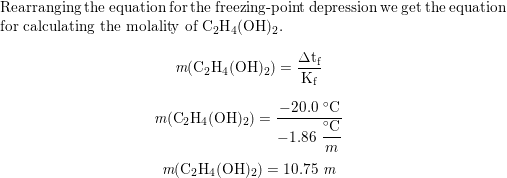
How many grams of antifreeze mathrm C 2 ma Quizlet
Ethylene oxide Wikipedia
10.3 Phase Change Diagrams Chemistry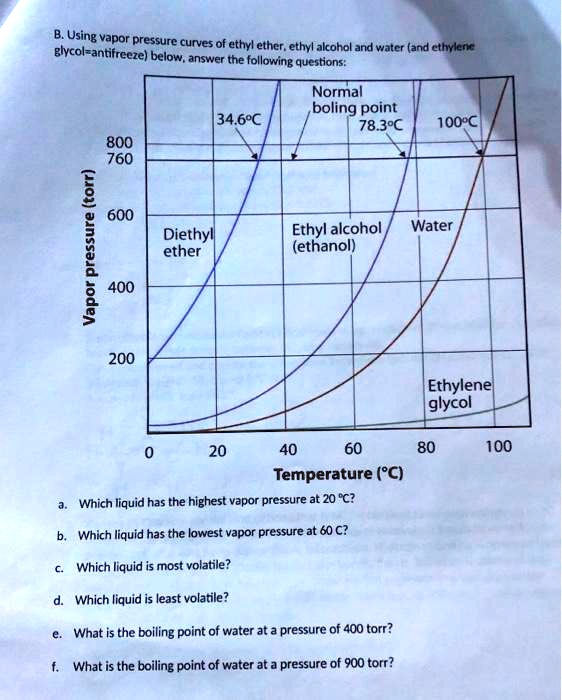
SOLVED Examining vapor pressure curves of ethyl ether ethyl
Plot a vapor pressure curve for C2H4F2 from the following vapor
SOLVED The vapor pressure of pure water at 60 C is 149 torr
Ethylene Density and Specific Weight vs. Temperature and Pressure
Ethylene Glycol CH2OHCH2OH CID 174 PubChem
Ethylene glycol C2H6O2 ChemSpider
Solved The vapor pressure of water at 20 C is 17.5 torr. A
What is the vapour pressure of a saturated solution of Ba OH 2 in
Ethane Density and Specific Weight vs. Temperature and Pressure
Answered 20 40 60 80 100 120 Temperature C bartleby
Simultaneously measurements of vapor pressure saturated liquid
ETHYLENE OXIDE PRODUCT STEWARDSHIP GUIDANCE MANUAL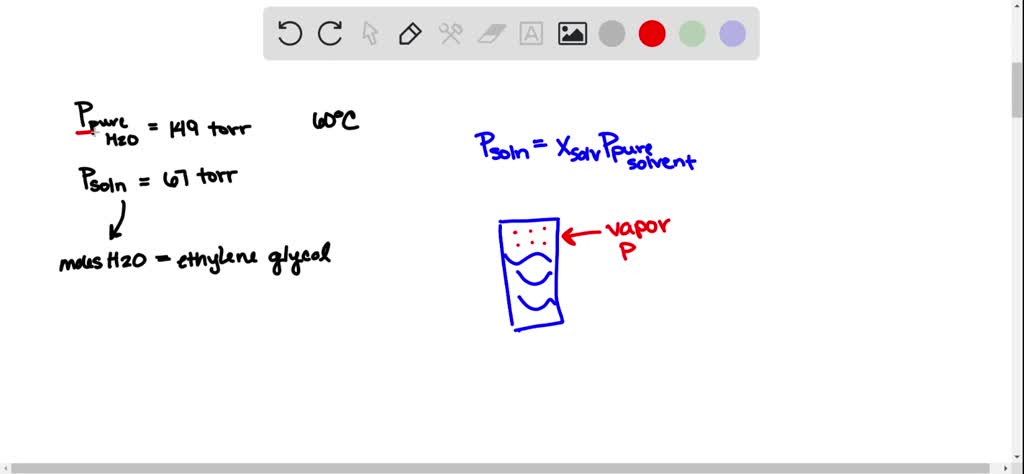
SOLVED The vapor pressure of pure water at 60 C is 149 torr
How many grams of ethylene glycol C2H4 OH 2 must be added to
Ethane Density and Specific Weight vs. Temperature and Pressure
Plot a vapor pressure curve for C2H4F2 from the following vapor
The 2 value as a function of the c C2H4O column density N and
Ethylene Glycol CH2OHCH2OH CID 174 PubChem
Ethylene Thermophysical Properties
Answered The graph shows the four common bartleby
Ethylene Dynamic and Kinematic Viscosity vs. Temperature and
The vapor pressure of pure water at 60 C is 149 torr. The vapor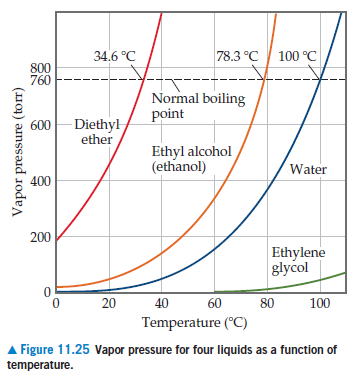
Answered 34.6 C 78.3 C 100 C 800 760 Normal bartleby
Chemistry Free Full Text Platform Chemicals from Ethylene
10.4 Phase Transitions Chemistry LibreTexts
A Fourier transform infrared spectra of neat ethylene glycol