TCV for CH4 and C2H4 and TCV T CH 4 CV T C 2 H 4 CV as a
Ch4 vs hot sale c2h4 boiling point
Share. Visit »
In each pair of compounds pick the one with the higher boiling p
Rank the following from lowest to highest anticipated boiling
Ethylene C2H4 Structure Molecular Mass Physical and Chemical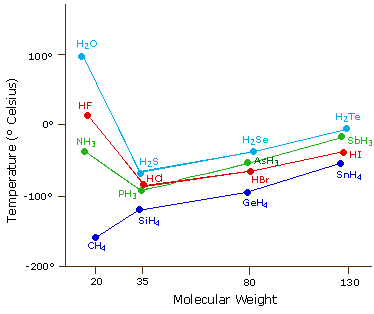
Supplemental Topics
Normal Boiling Point C1 C5 PDF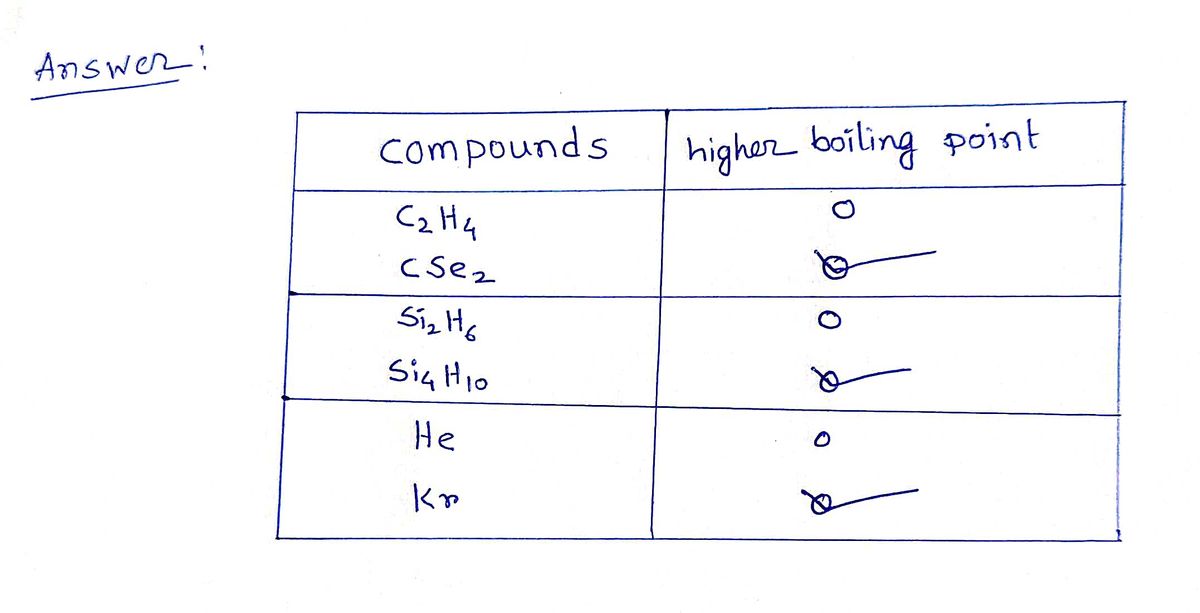
Answered For each pair of compounds listed bartleby
Which one has the highest boiling point methanol or methane Quora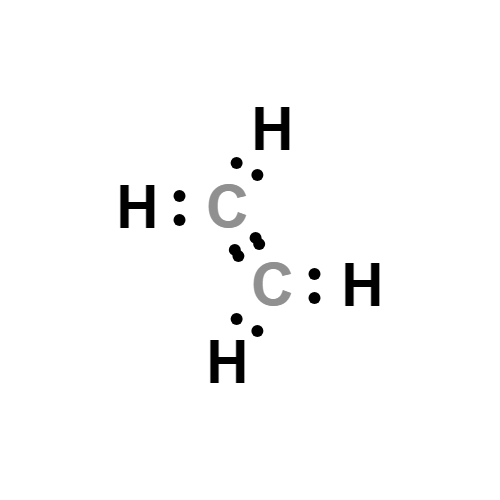
ETHYLENE 74 85 1
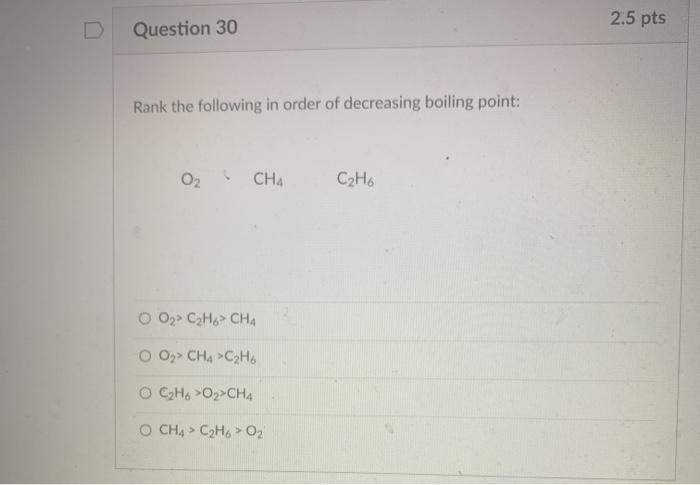
Solved 2.5 pts Question 30 Rank the following in order of Chegg
Ripening Ethylene C2h4 Gas China Gas Cylinder Cylinder Made
In each pair of compounds pick the one with the higher boiling p
Ethylene Thermophysical Properties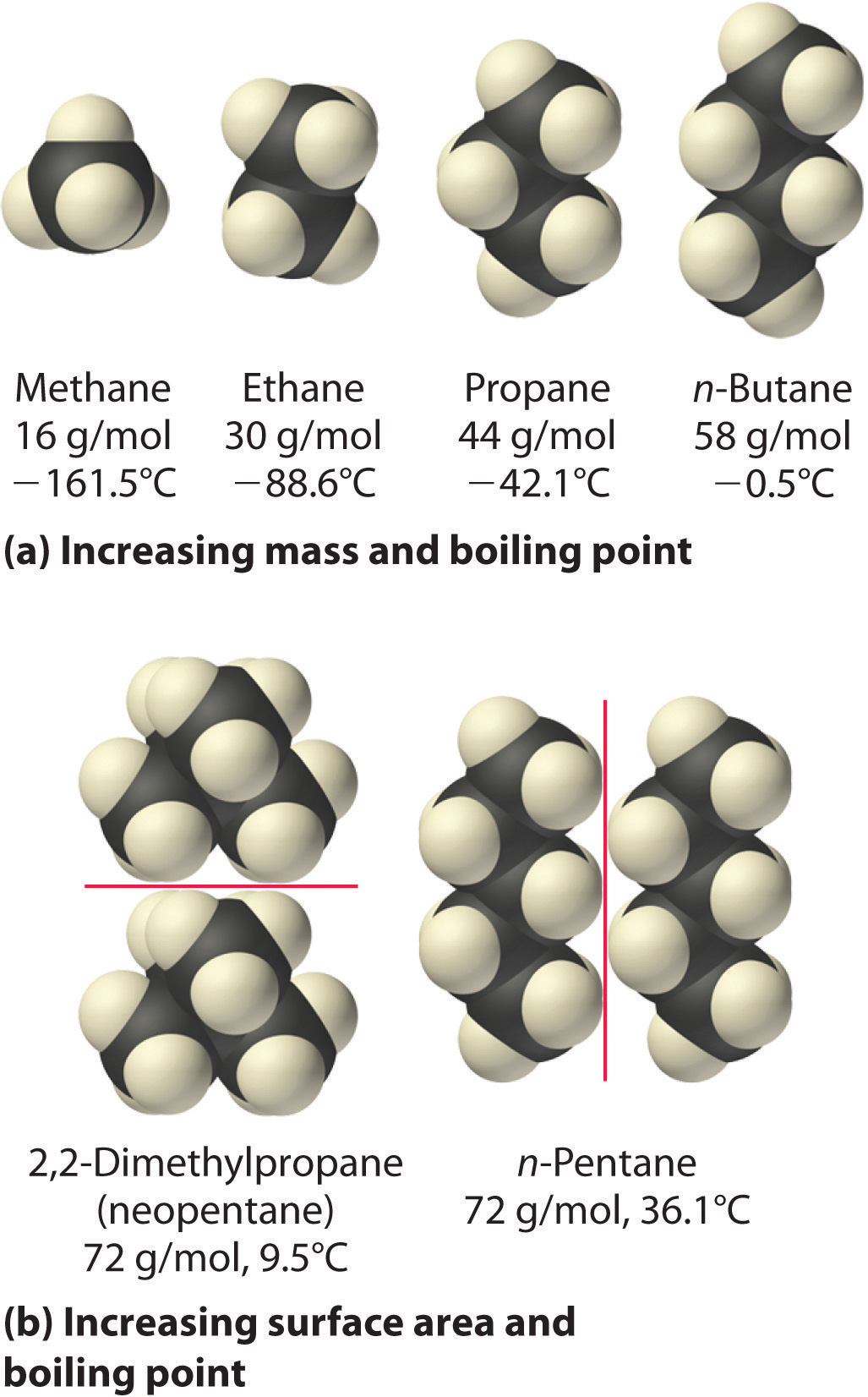
Which molecule would have the largest dispersion molecules forces
Adsorption isotherms of different gas species CH4 C2H6 C2H4
Difference in Boiling Point for CH4 and CCl4 Methane and Carbon
Answered .Methane molecule CH4 hybridization is bartleby
Topic 8 Case study polyethylene high impact polystyrene ppt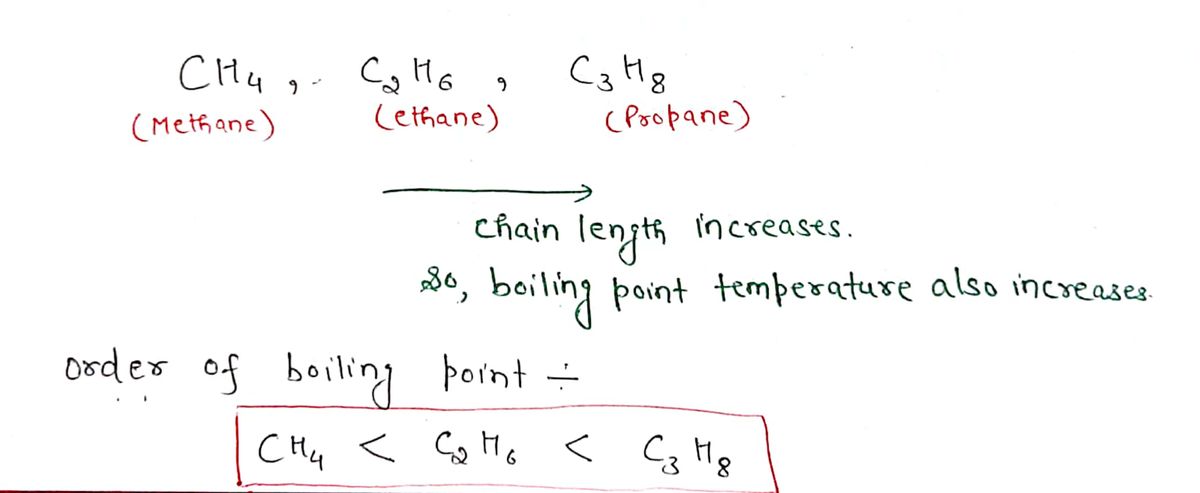
Answered Arrange CH4 C2H6 C3H3in order of bartleby
C2H4 Specialty Gas C2H4 Gas
Answers PDF
Boiling Point of Methane CH4
Consider the following CH4 SiH4 GeH4 SnH4 The boiling points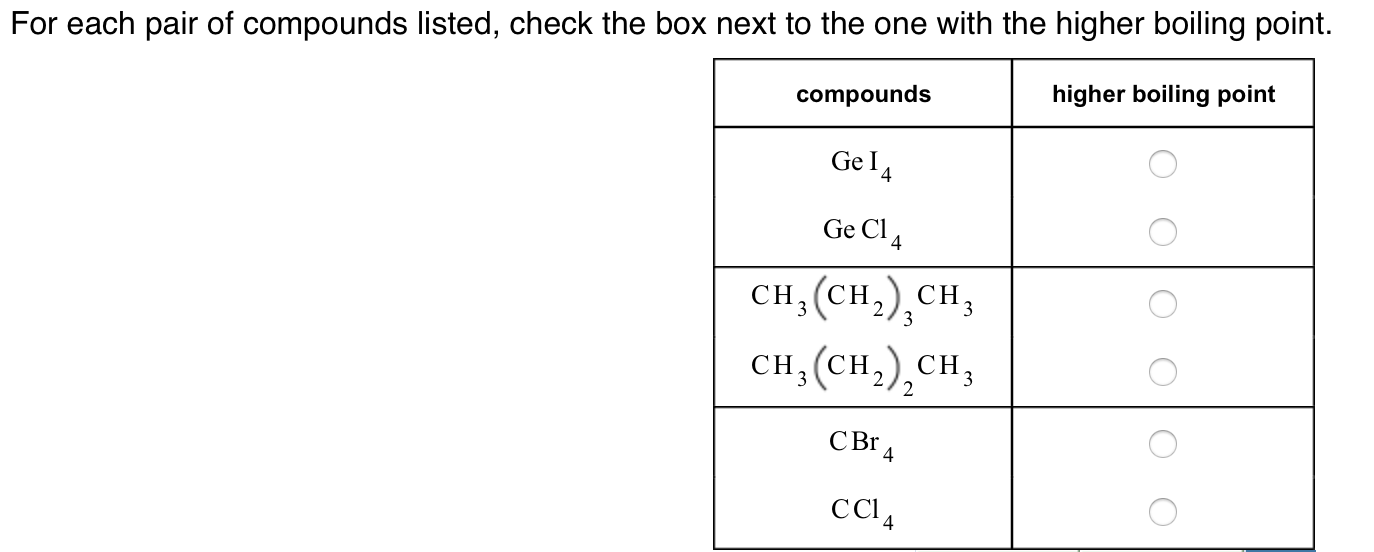
Solved For each pair of compounds listed check the box next
Hf of c2H2 c2H4 c2H6 respectively are 226 56.the difference
SOLVED Which will have the highest boiling point. N2 HCl CO2 Ne
Ethylene Wikipedia
Ethylene Wikipedia
Solved Which of the following compounds would be most likely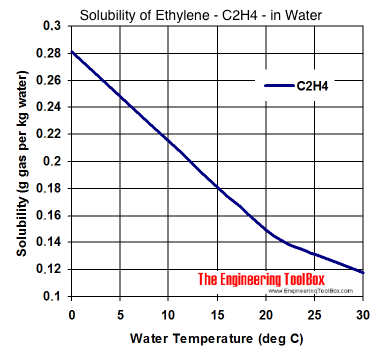
Solubility of Gases in Water vs. Temperature
Solved Place these compounds in order from lowest boiling
Highly permeable ZIF 8 membranes for C2H4 C2H6 separation in a
Difference in Boiling Point for CH4 and CCl4 Methane and Carbon
Ethylene CH2 CH2 CID 6325 PubChem
SOLVED For each pair of compounds listed check the box next to
Solved Which of the following has the highest boiling point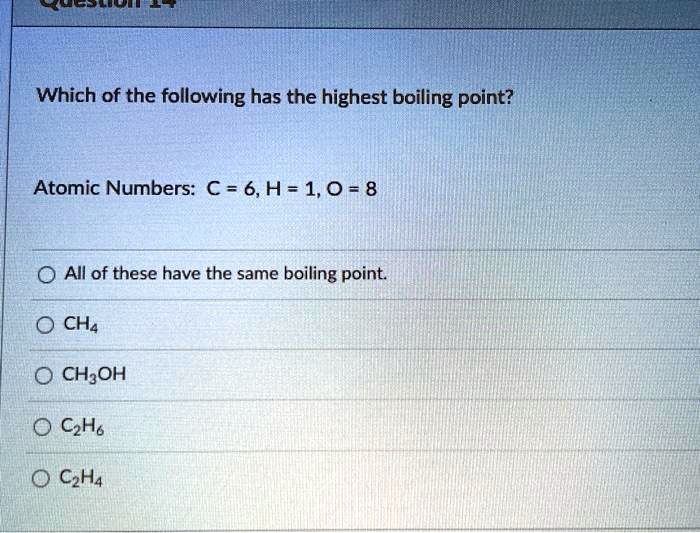
SOLVED Which of the following has the highest boiling point
WO2006017579A2 High energy transport gas and method to transport
Ethylene Density and Specific Weight vs. Temperature and Pressure
Arrange these compounds in order of increasing boiling point