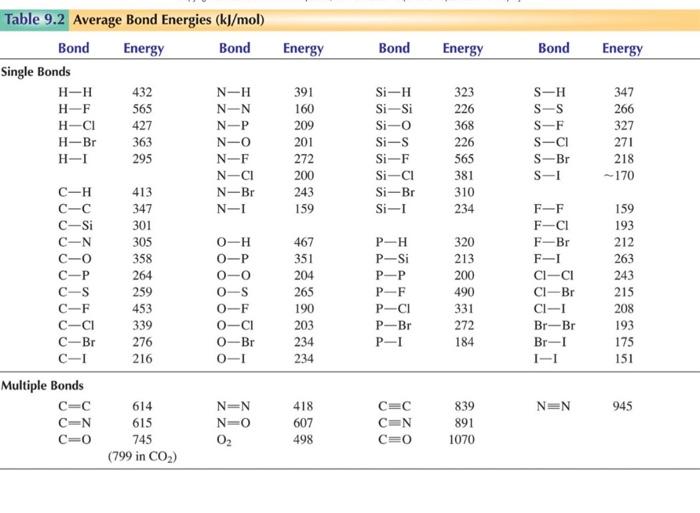
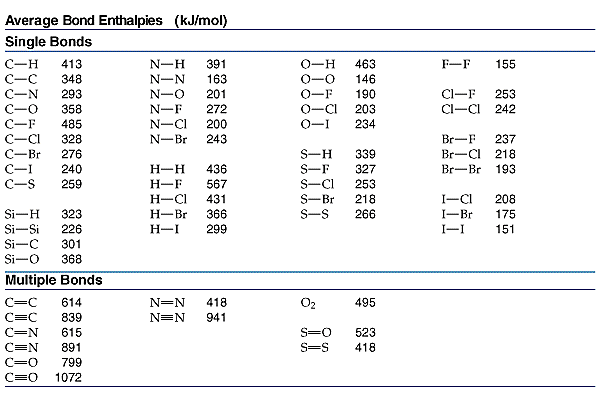
Use bond energies to confirm that the complete combustion of
Energy to break hot sale c2h4
Share. Visit »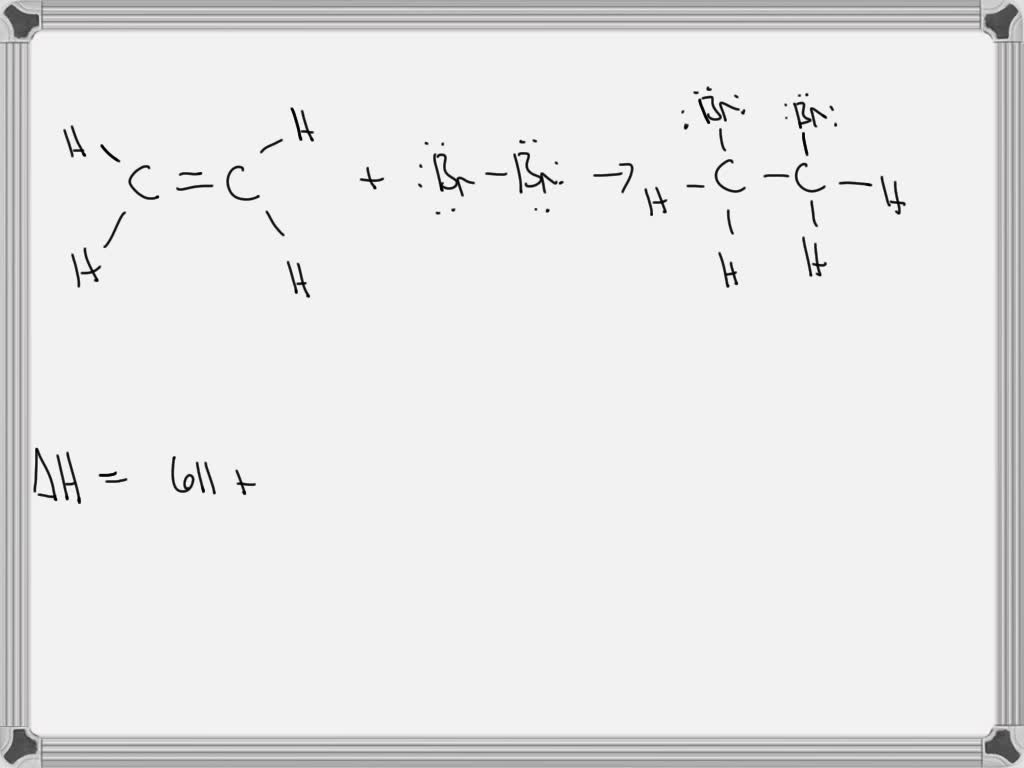
SOLVED Consider the bromination of ethene C2H4 g Br2 g
Using the bond enthalpy data given below calculate the enthalpy
Enthalpies of Reactions Using Average Bond Enthalpies Chemistry Tutorial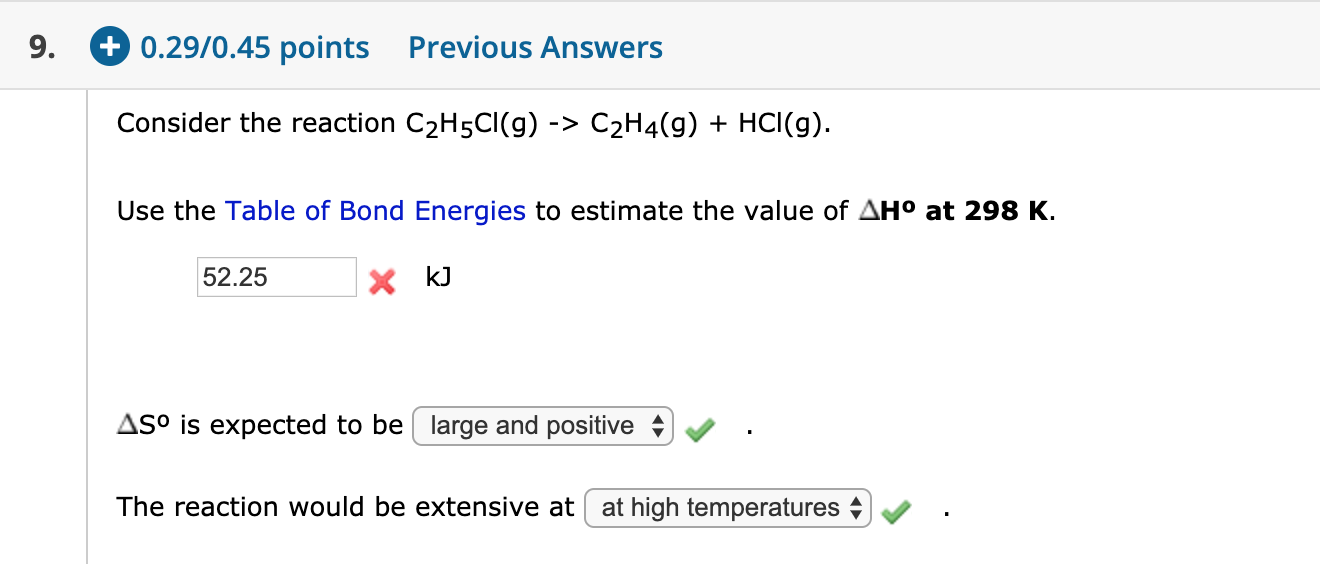
Solved 9. 0.29 0.45 points Previous Answers Consider the Chegg
Determination of the Rh C bond energy for C2H2 and C2H4 reactive
Bond Enthalpy Table Equation Calculations Video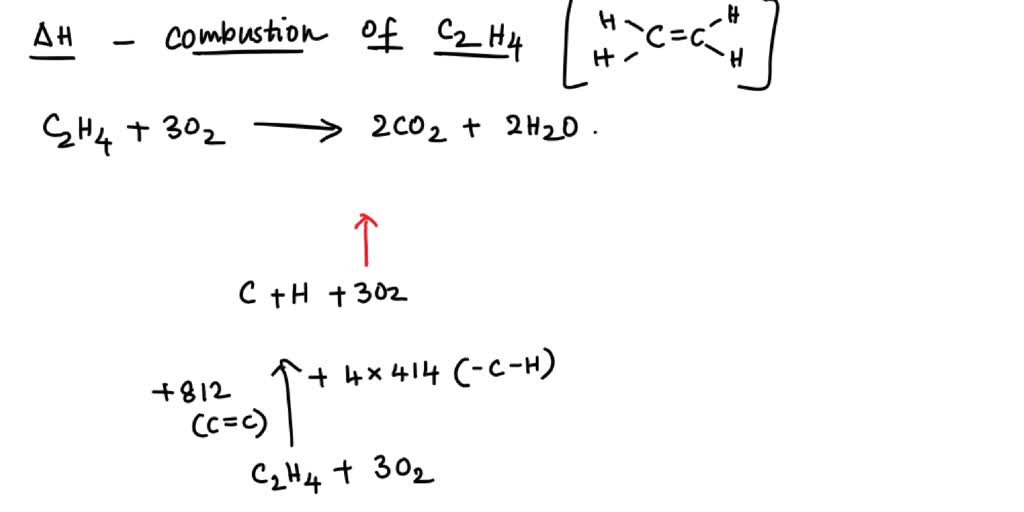
SOLVED Using the bond enthalpy values provided calculate the
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol
Understanding Hess Law C2H4 H2O C2H5OH
Chapter 9 Reaction Energetics
SOLVED Calculate the enthalpy change for the reaction of ethene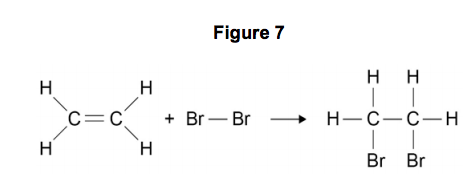
G A C H P1 S1 Q9 A Elevise
Solved Question 30 1 point Use the bond energies provided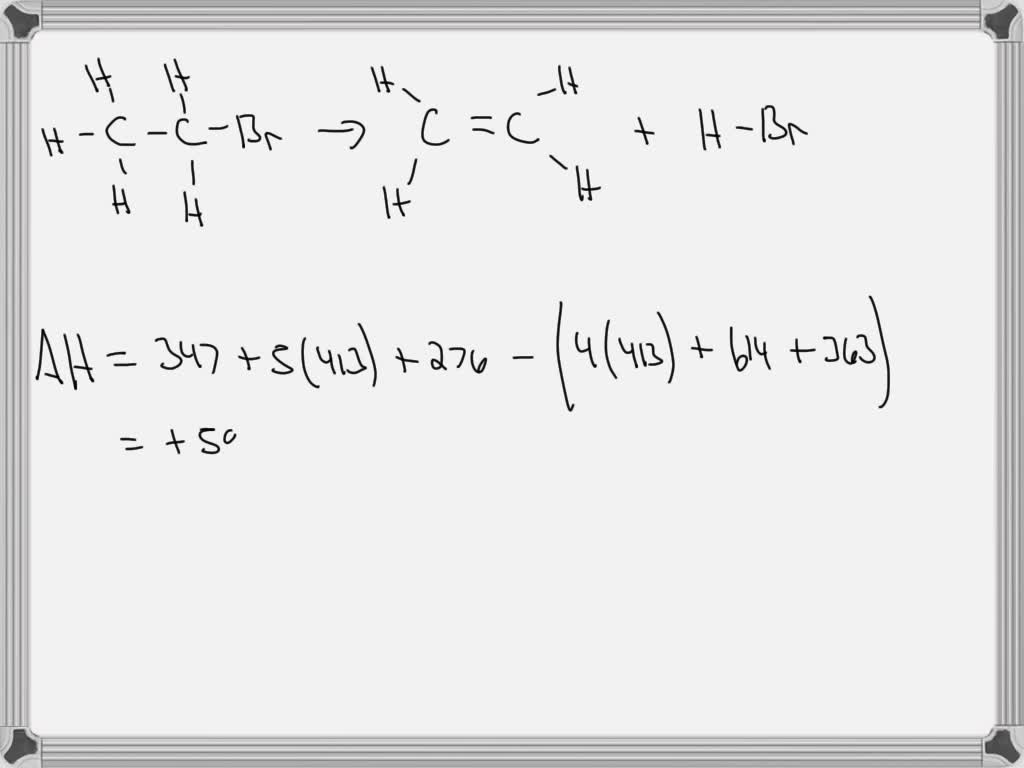
SOLVED Consider the following reaction C2H5Br g C2H4 g
What is the heat in kJ mol released during the gas phase
Bond Energy Worksheets by Olivia Hunter Issuu
Using bond enthalpies to calculate an enthalpy change of reaction
Bond Enthalpies
18. Calculate the enthalpy of hydrogenation of C2H2 g to C2H4 g
Solved Use Lewis structures and bond energies to calculate Chegg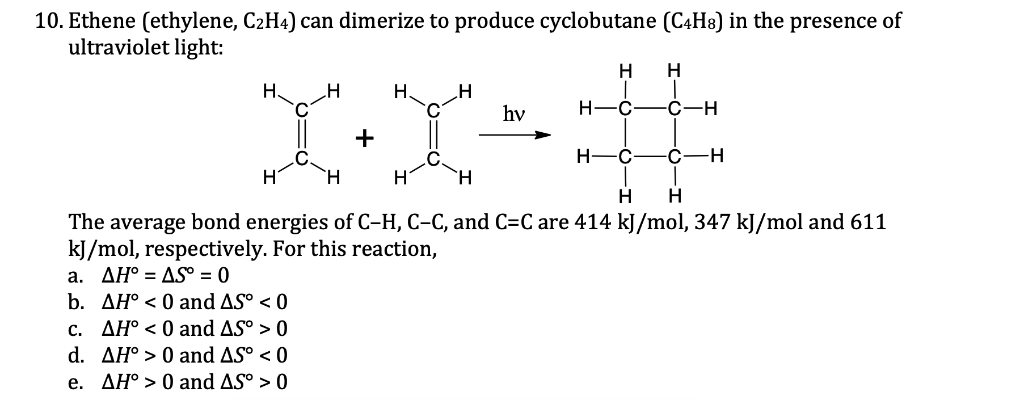
Solved 10. Ethene ethylene C2H4 can dimerize to produce Chegg
Calculating the Carbon Iodine Bond Energy in Iodination of Ethene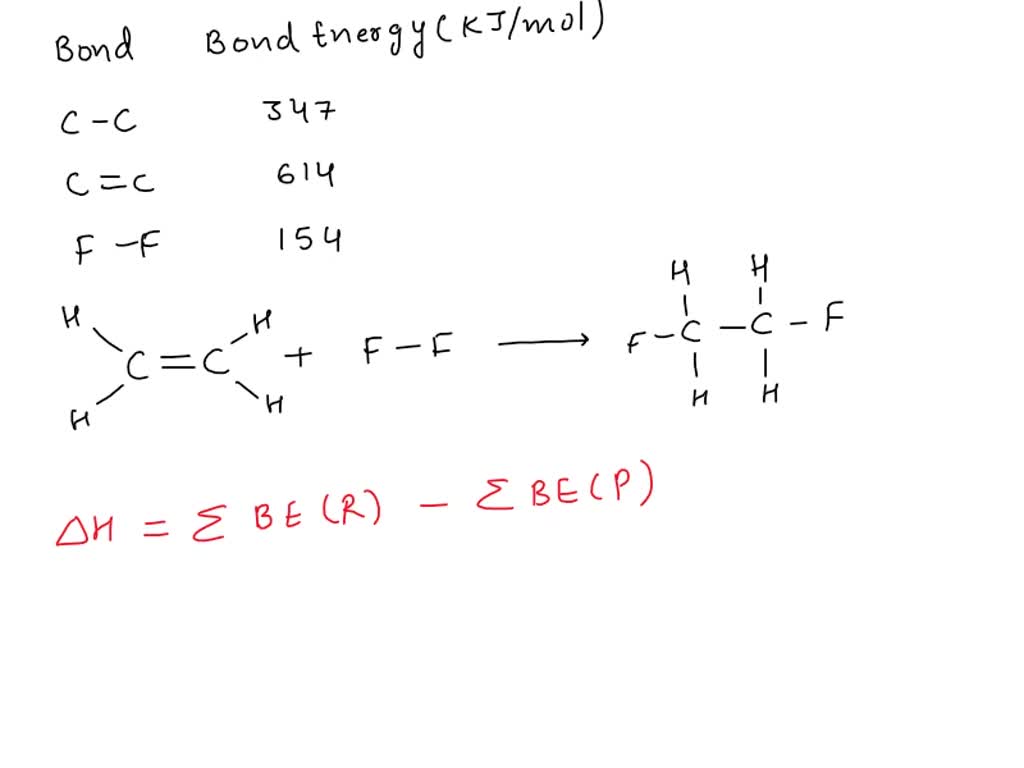
SOLVED Consider the following reaction C2H4 g F2 g
Calculating the Molar Bond Energy for C H Bonds Given the Amount of Energy Required to Break All C H Bonds in Methane
Ethylene Wikipedia
The enthalpy change for the following reaction is 137 kJ. Using
The C2H4 C2F4 charge transfer energy in different intermolecular
Chemical Energy 07 12 ppt download
Enthalpy. ppt download
Solved Problem 5. Energetics of Chemical Bonds The Chegg H2(g) → C2H6(g).jpg)
1.2.6 Bond Enthalpies. ppt download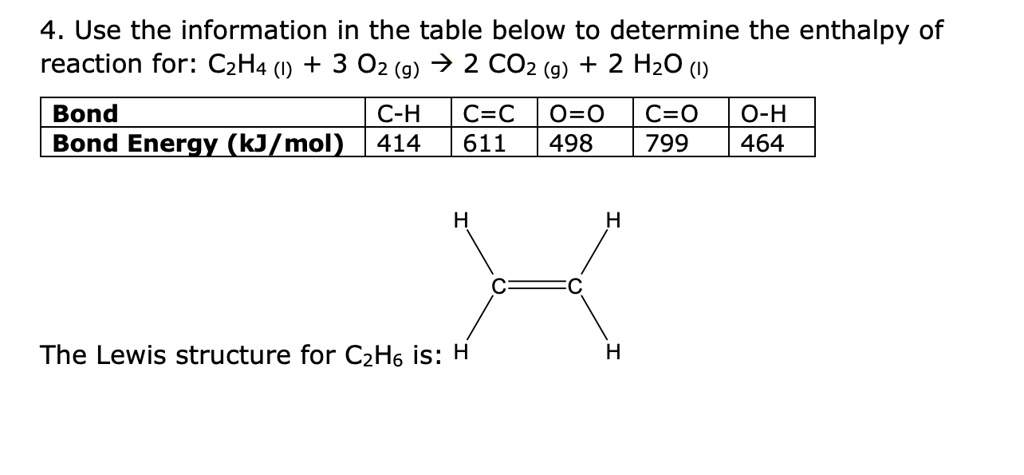
SOLVED Use the information in the table below to determine the
Comparison of the selectivity energy efficiency and electric
Bond Enthalpy Bond Energy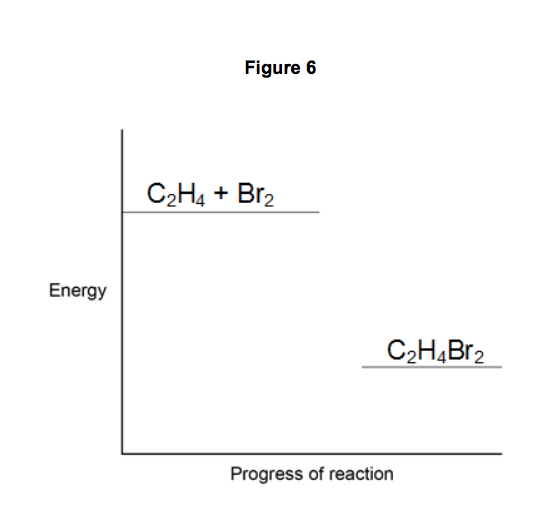
G A C H P1 S1 Q9 A Elevise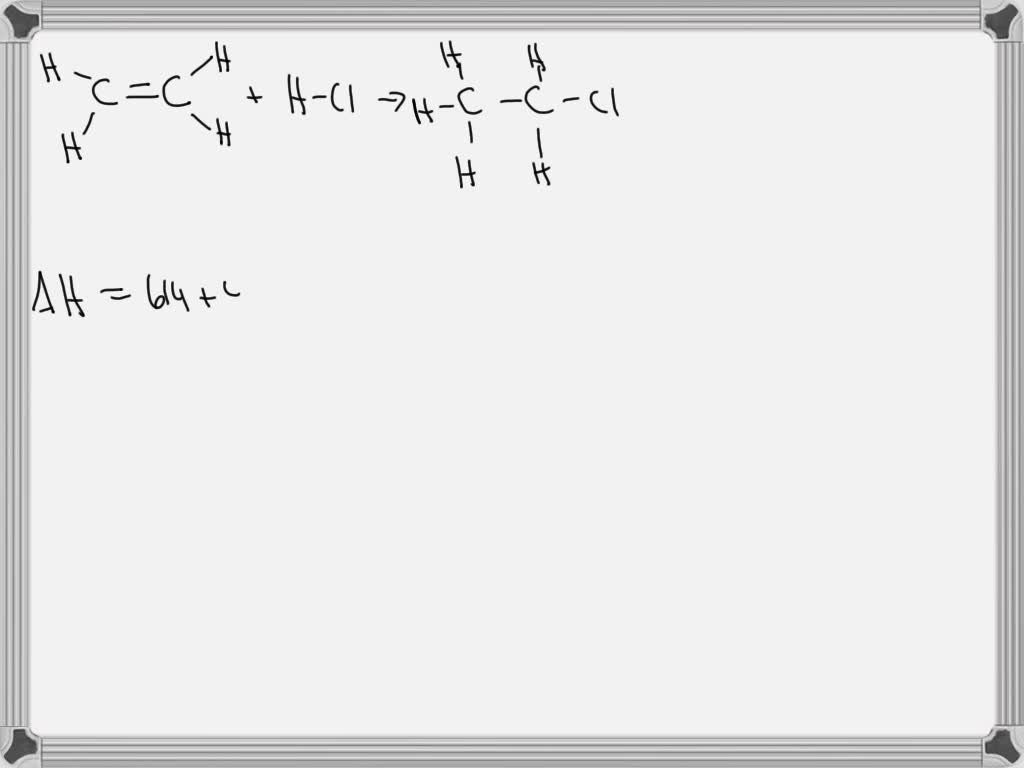
SOLVED Using the table of bond dissociation energies the H for
Bond Enthalpy Bond Energy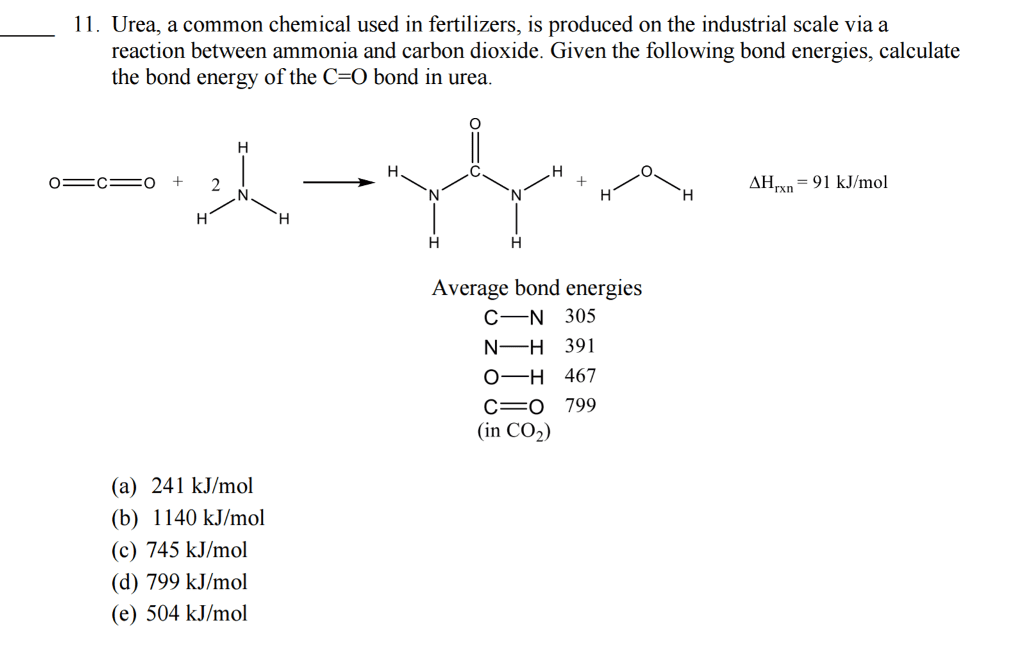
OneClass Urea a common chemical used in fertilizers is produced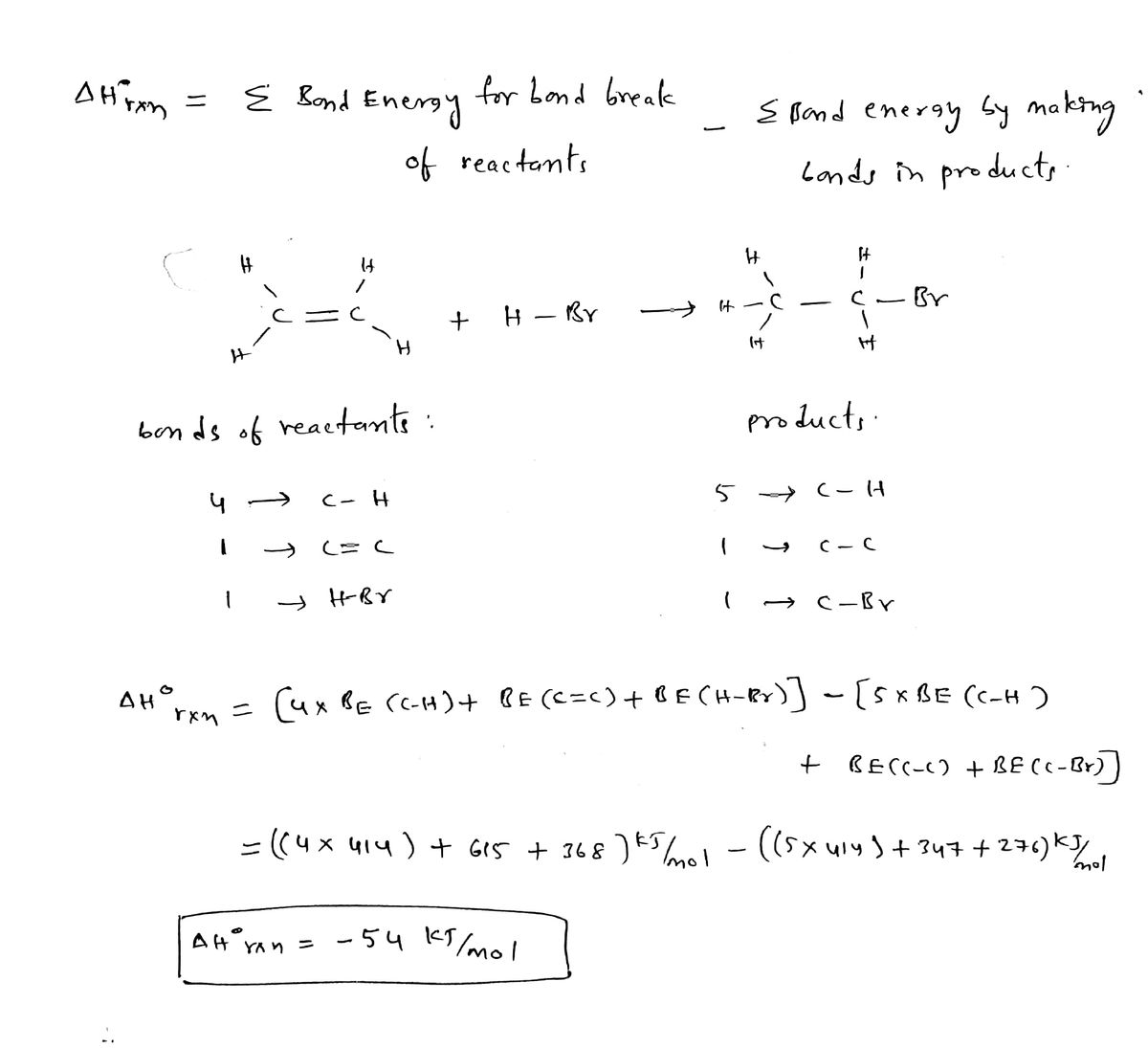
Answered Use the molar bond enthalpy data in the bartleby