C2H6 C2H4 H2. If the enthalpy change for this reaction is 137
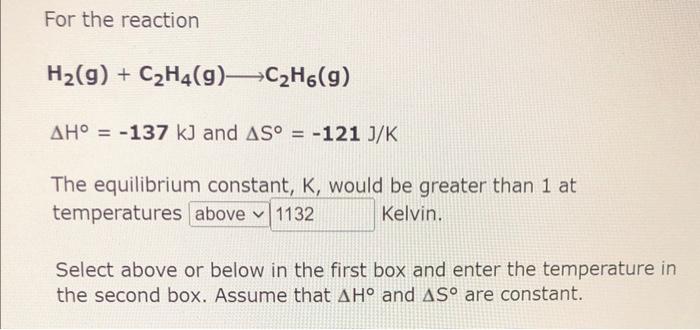
For the reaction hot sale c2h6 137kj c2h4 h2
Share. Visit »
For the reaction C2H4 H2 C2H6 E 30 kcal
SOLUTION Chm1020 week6 docx Studypool
Solved For the reaction H2 g C2H4 g C2H6 g H 137 kJ
How to Balance C2H2 H2 C2H6 Hydrogenation of Acetylene
Week 6 Assignment.pdf Week 6 Assignment Multiple Choice 1 The
Answered Consider the reaction below at 25 C 2 bartleby
SOLVED Texts For the reaction C2H6 g C2H4 g H2 g H
The enthalpy change for the following reaction is 137 kJ. C2H4 g
18. Identify oxidation and reduction process C2H4 H2 C2H6
Hess Law Learning Goals ppt download
Chemistry 4.4 Hess s law Flashcards Quizlet
OneClass For the reaction C2H6 g C2H4 g H2 g Delta
Solved 24 For the reaction C2H6 g C2H4 g H2 g AH
THERMOCHEMISTRY Thermodynamics ppt download
SOLUTION Palomeras marvin g bsee 1 1 assignment lesson 5 unit 03
SOLVED For the reaction C2H6 g C2H4 g H2 g H is
C2H6 C2H4 H2. If the enthalpy change for this reaction is 137
Reaction mechanism of transformation of C2H6 to C2H4 over VOx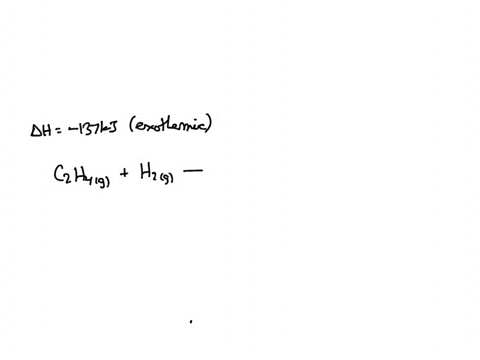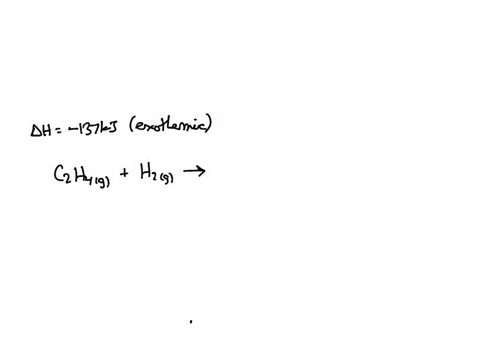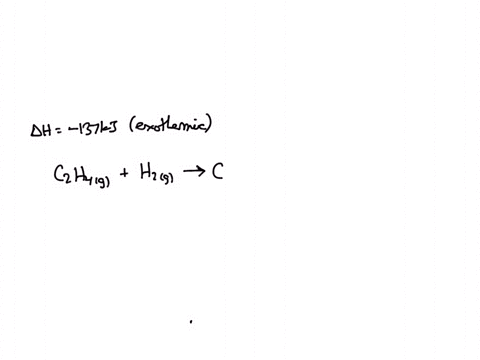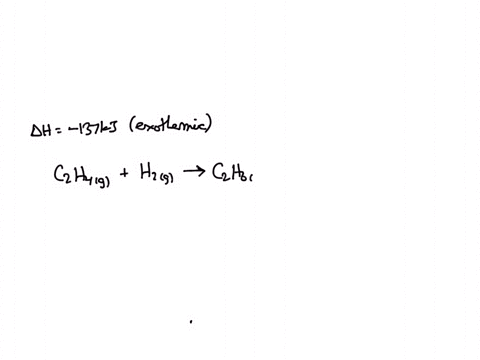
SOLVED Consider the reaction C2H4 g H2 g C2H6 g where
Answered 3D 13 Consider the reaction C2H4 g bartleby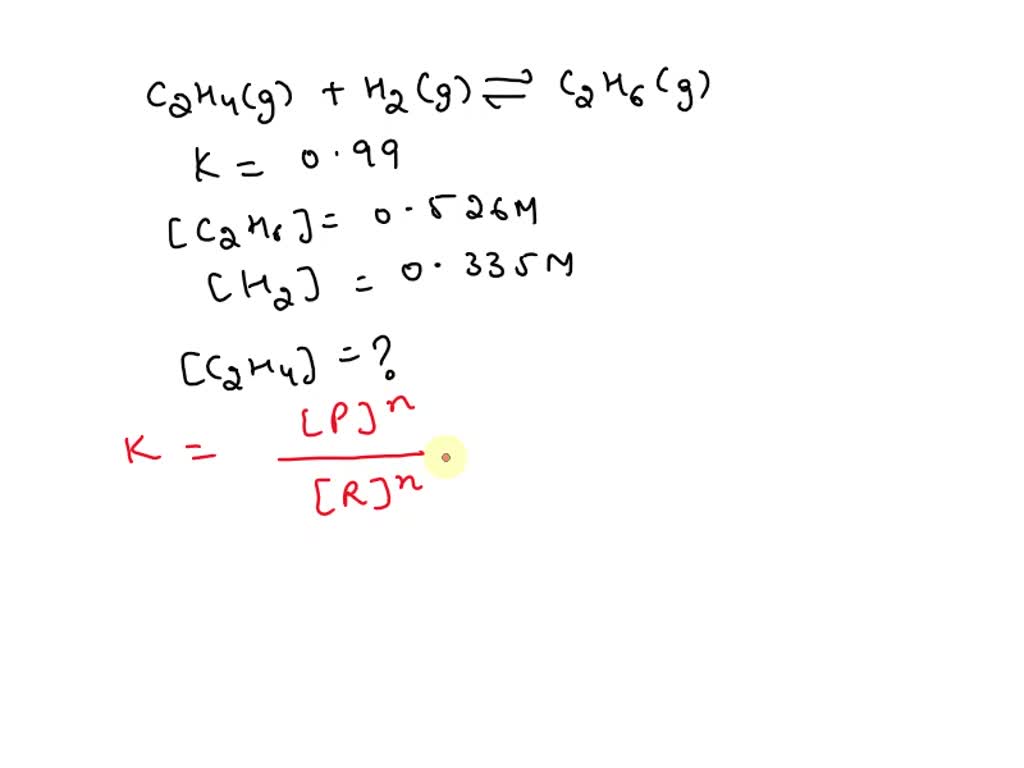
SOLVED Consider the following balanced reaction C2H4 g H2 g
Components of the Heats of Reactions SpringerLink
The enthalpy change for the following reaction is 137 kJ. Using
Elementary steps for the reaction C2H2 H2 C2H4 and C2H4 H2
Calculating Standard Enthalpy Changes from Thermochemical Data
Solved Consider the reaction C2H6 g C2H4 g H2 g For
PPT 2 AlBr 3 3 Cl 2 PowerPoint Presentation free download
Solved For the reaction C2H6 g rightarrow C2H4 g H2 g
Hess s law PPT
Chemistry 30
Solved For the reaction C2H4 g H2 g C2H6 g H is
Solved 39. For the reaction C2H6 g C2H4 g H2 g AH
OneClass For the reaction C2H6 g C2H4 g H2 g Delta
C2H4 h2 c2h6is which type of reaction Quora
Solved Consider the reaction C2H4 g H2 g C2H6 g where
Solved For the reaction C2H6 g C2H4 g H2 g AH rxn
Consider the reaction C 2 H 4 g H 2 g C 2 H 6 g where H 137 kJ
C2H4 g H2 g C2H6 g H 137.5 kJ S 120.5 J K
The enthalpy change for the following reaction is 137 kJ. Using