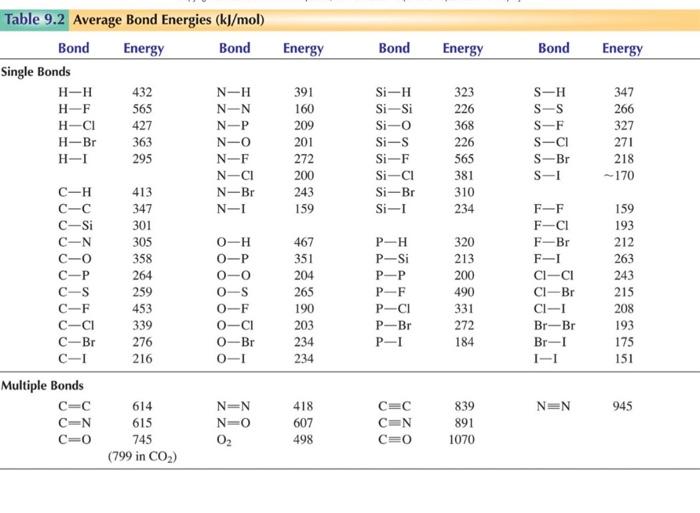
How to use the average bond dissociation energies in the table to
Bond dissociation energy between 2ch4 g c2h4 hot sale g 2h2 g
Share. Visit »
Bond Energy and Reaction Enthalpy ppt download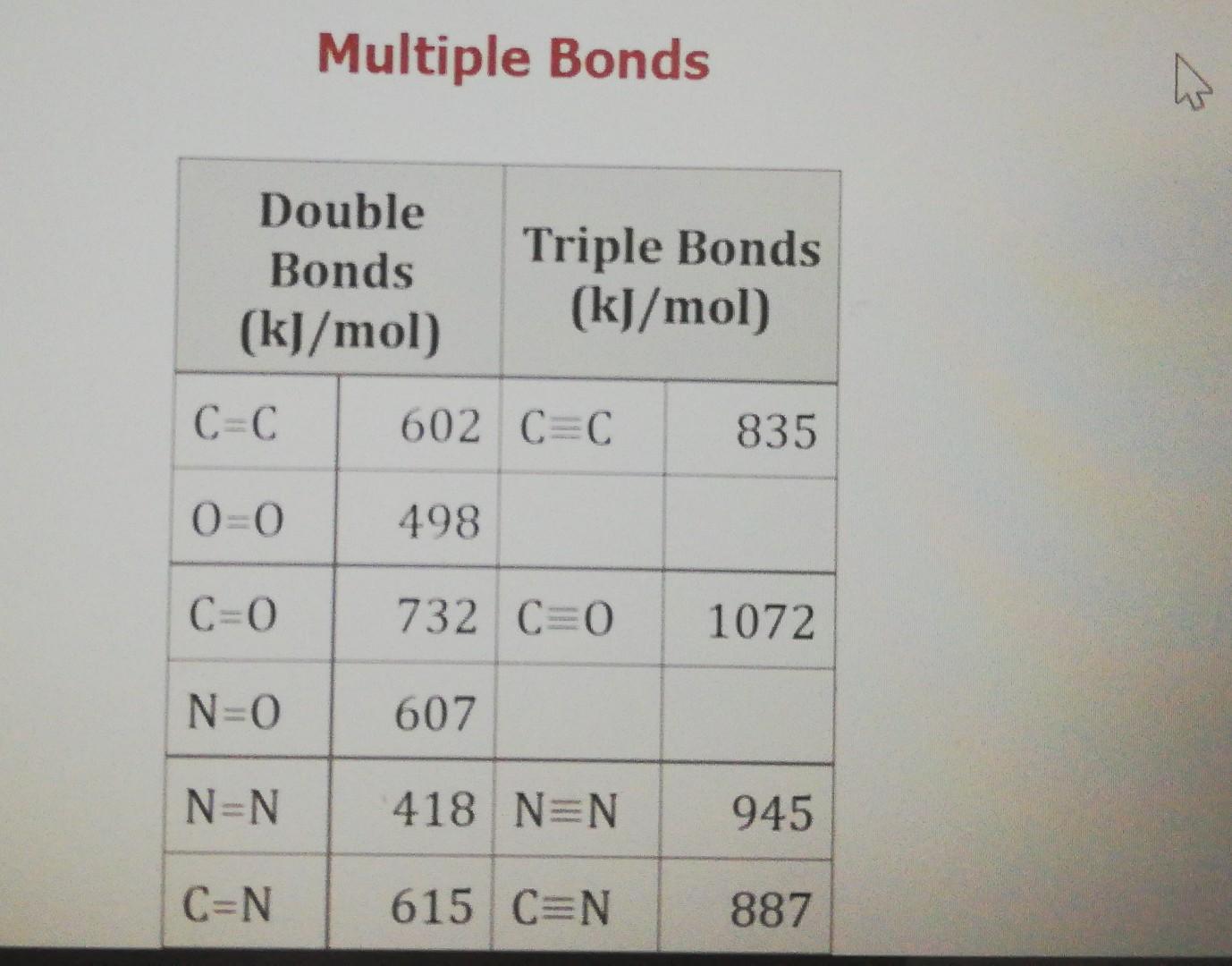
Solved Using the values of bond energy from the table below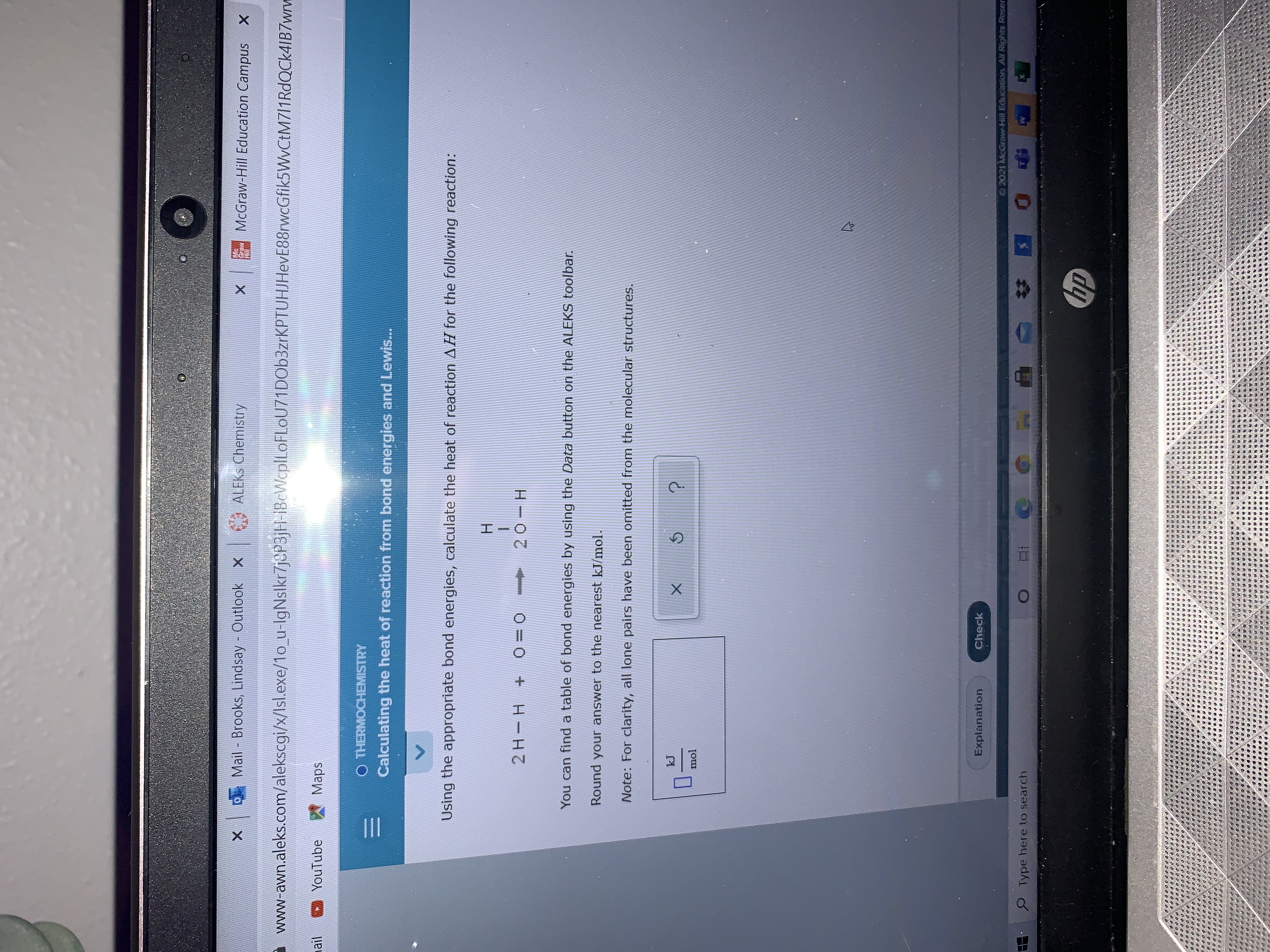
Answered Using the appropriate bond energies bartleby
What is the difference between bond dissociation enthalpy and
Solved Calculate AH for the reaction using the given bond Chegg
Solved Determine the enthalpy of reaction for 2CH4 g Chegg
What is the difference between bond dissociation enthalpy and
Using Bond Energy to find Enthalphy 2O2(g) 2H2O(l) CO2(g).jpg)
Objectives understand that chemical reactions involve the making
SOLVED 2CH4 g C2H4 g 2H2 g 2CH4 g CH4 g 2H2 g a
Answered Use the bond enthalpies given below to bartleby
Solved Calculate Delta H for the following reaction using Chegg
Use bond energies to confirm that the complete combustion of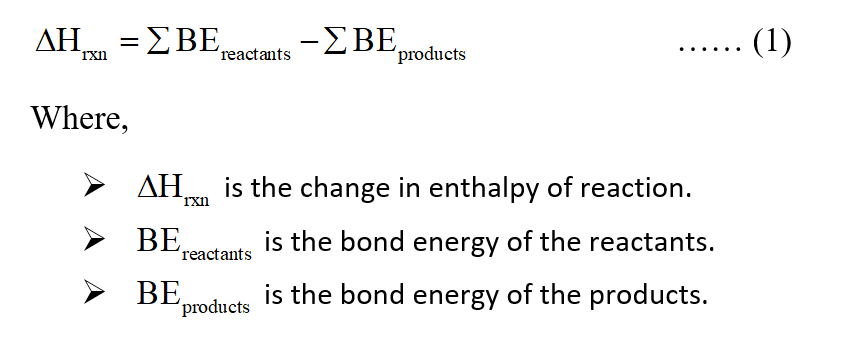
Answered Use the bond enthalpies given below to bartleby 2O2(g) 2H2O(l) CO2(g).jpg)
THERMOCHEMISTRY Thermodynamics ppt download
7.65a How to calculate the enthalpy change from bond energies
SOLVED 2CH4 g C2H4 g 2H2 g 2CH4 g CH4 g 2H2 g a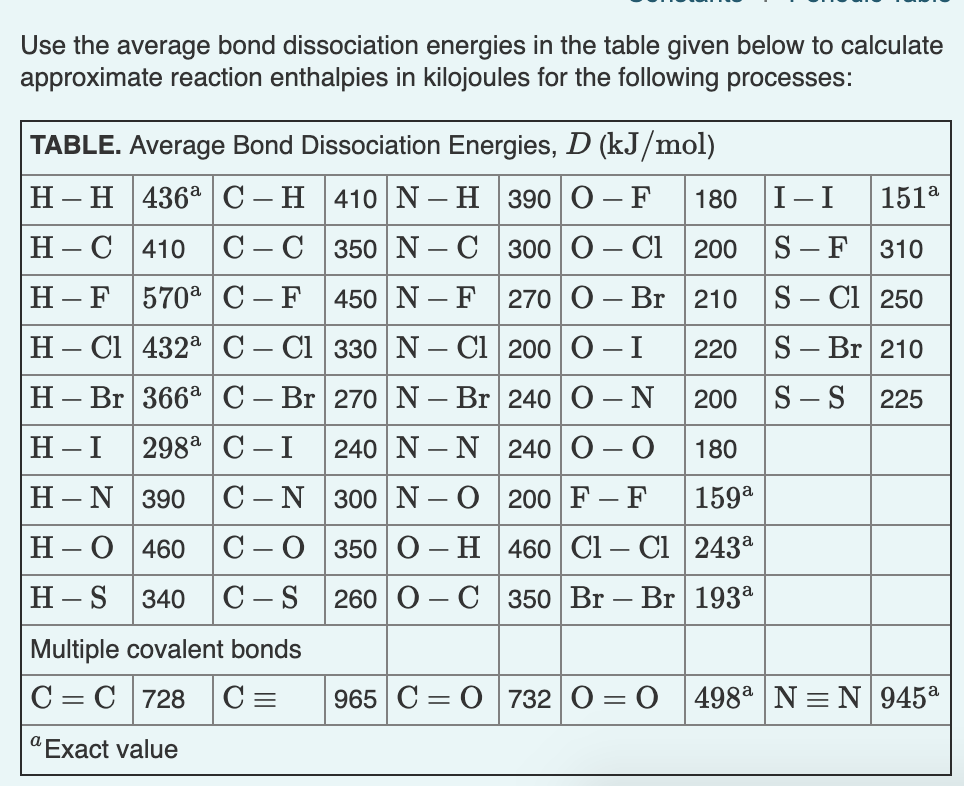
Solved 2CH4 g C2H6 g H2 g Chegg
IB Chemistry on Hess s Law Enthalpy Formation and Combustion PPT
Answered I. AHxn for the reaction below was bartleby
Solved Use Lewis structures and bond energies to calculate Chegg
Answered Using the table of bond dissociation bartleby
How to use the average bond dissociation energies in the table to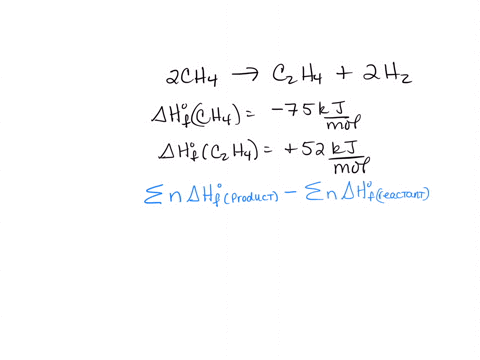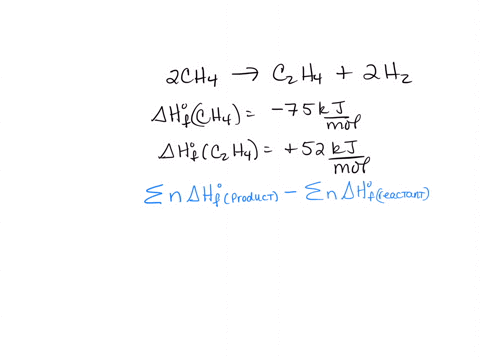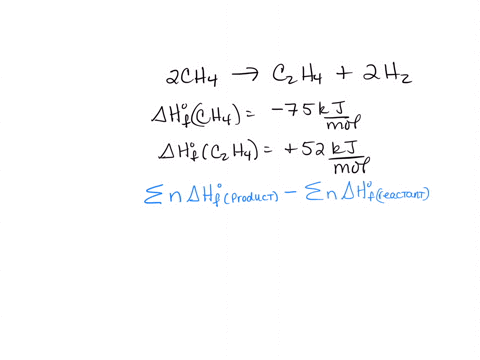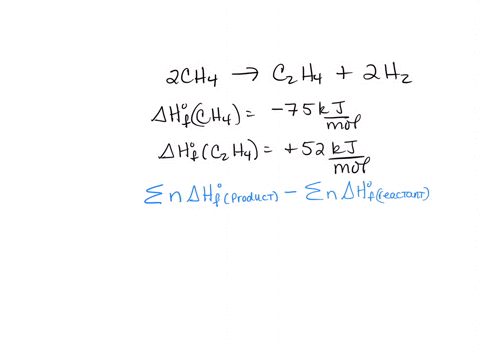
SOLVED 2CH4 g C2H4 g 2H2 g 2CH4 g CH4 g 2H2 g a
Solved . Calculate for the reaction C2H2 g 2 H2 g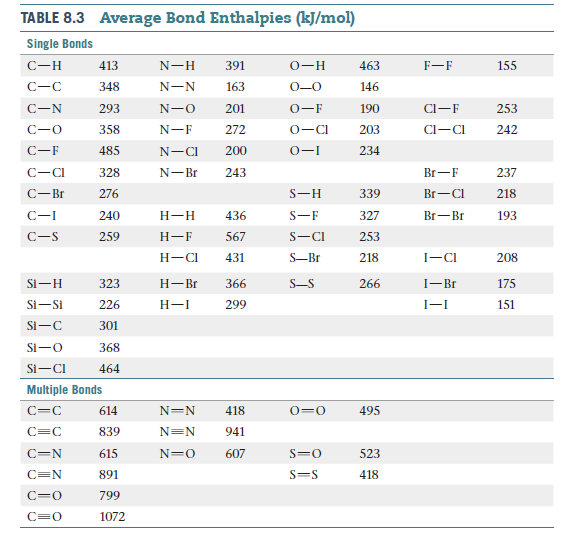
Answered TABLE 8.3 Average Bond Enthalpies bartleby
Markscheme 63 Marks PDF
IB Chemistry on Hess s Law Enthalpy Formation and Combustion PPT
The enthalpy change for the reaction 4 H g 2 H 2 g is
Answered I. AHxn for the reaction below was bartleby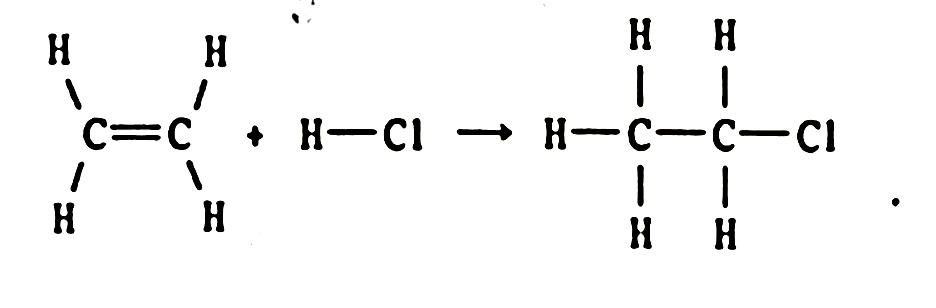
Answered Using the table of bond dissociation bartleby
The enthalpy change for the reaction 4 H g 2 H 2 g is 869.6 kJ. The dissociation energy of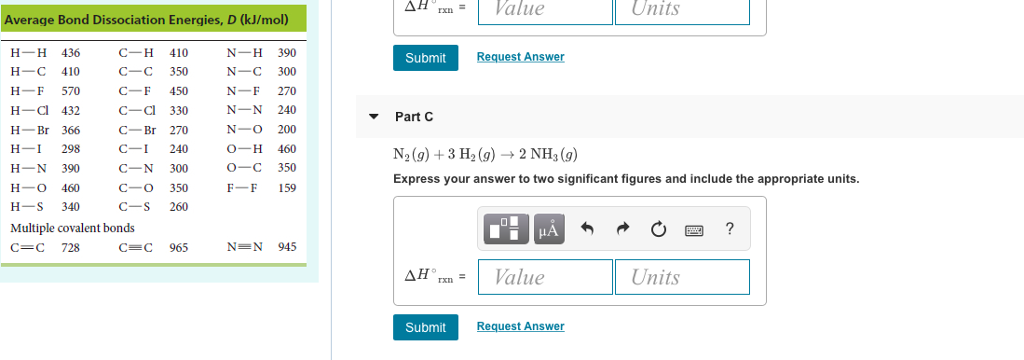
Solved ConstantsI Part A Use the average bond dissociation
How to use the average bond dissociation energies in the table to
What is the difference between bond dissociation enthalpy and
SOLVED CH4 g 2 O2 g CO2 g 2 H2O g H2O Which of the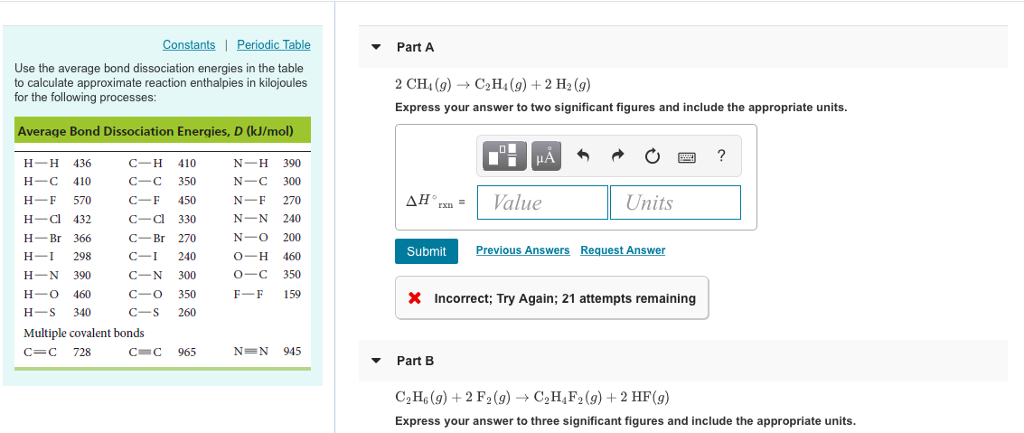
Solved ConstantsI Part A Use the average bond dissociation
Solved 4. Use bond dissociation energies to calculate the Chegg
18. Calculate the enthalpy of hydrogenation of C2H2 g to C2H4 g