calculate the heat of combustion of ethene Brainly.in
Calculate the heat of combustion of ethylene hot sale c2h4
Share. Visit »
Calculate the ethalpy of combustion of ethylene g to form CO 2 gas and H 2 O gas at
Solved Use the equation to help you calculate the heat of Chegg
OneClass The combustion of ethane C2H4 occurs via the reaction
Answered For the combustion reaction of ethylene bartleby
SOLVED Given the heats of combustion of ethylene hydrogen and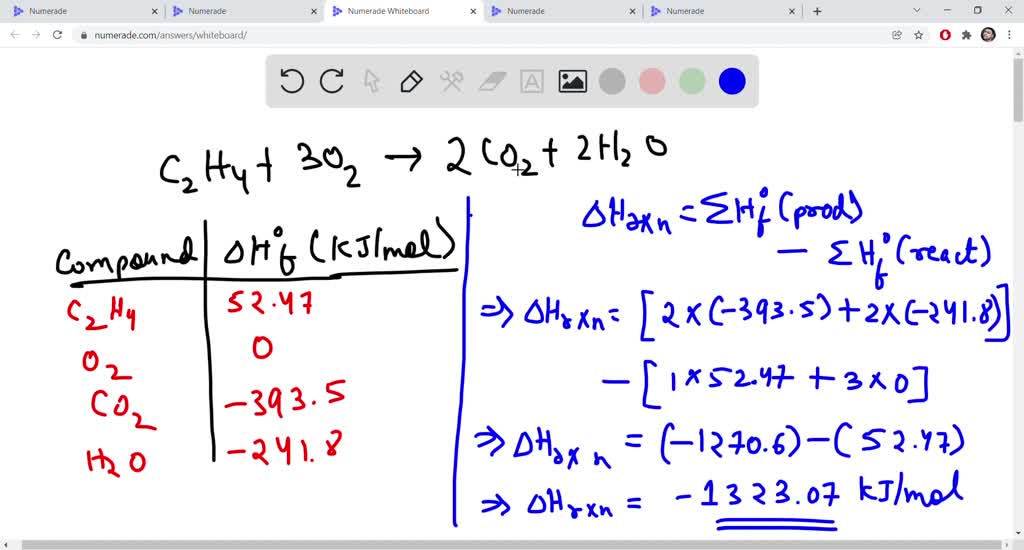
SOLVED The combustion of ethene C2H4 occurs via the reaction
42. Standard enthalpies of combustion of C2H4 g C2H6 g and H2 g
Calculate the ethalpy of combustion of ethylene g to form CO 2 gas and H 2 O gas at 298 K and 1 atmospheric pressure. The enthalpies of formation of CO 2 H 2 O and C 2 H 4 are
ntif enthalpies of formation for c2h4 g co2 g and h2o l at
Given the following standard heats of reactions heat of
Solved the standard enthalpy of formation of ethylene Chegg
Answered If the complete combustion of an bartleby
1. The heat of combustion of ethylene at 17 C and at constant
SOLVED The standard enthalpy of combustion of ethene gas C2 H4
5.7 Enthalpy Calculations Chemistry LibreTexts
The thermochemical equation for the combustion of ethylene gas C 2 H 4 is C 2 H 4 g
The heat of combustion of ethylene 17 C and constant volume is
Ethylene C2H4 is burned with 20 excess air in an adiabatic
Solved Use the equation to help you calculate the heat of Chegg
when a certain amount of ethylene c2h4 was combusted 6226 kJ
Calculate the enthalpy of combustion of ethylene 1 atm pressure
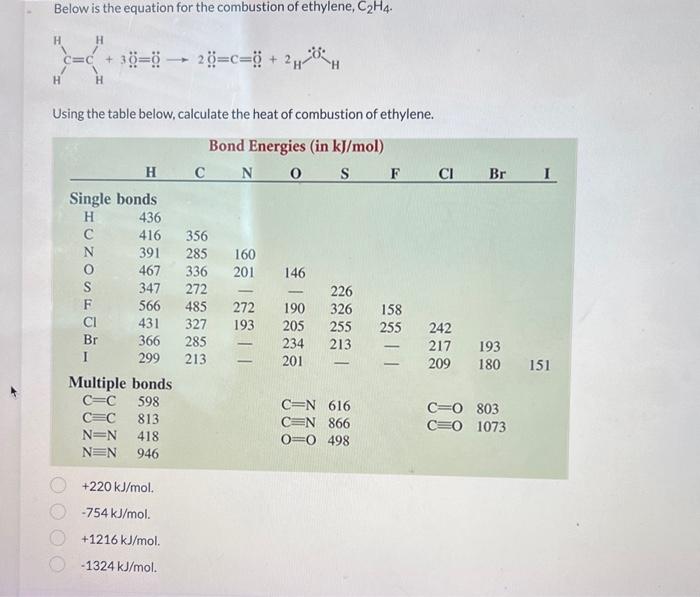
Solved Below is the equation for the combustion of ethylene
39 The heat of combustion of ethene C.H is 1409.3 kJ mol
Solved 3 Use the equation to help you calculate the heat of
Calculate the heat of combustion of ethylene to form co and H O
Solved 5 Calculate the standard heat for combustion of Chegg
Calculate the ethalpy of combustion of ethylene g to form CO 2 gas and H 2 O gas at
Ethylene on combustion gives carbon dioxide and water. Its heat of combustion
The heat of combustion of ethylene 17 C and constant volume is
31. Calculate the heat of combustion of ethene CH2 CH2 g 3O2 g
Consider the combustion of ethylene C2H4 3 O2 g 2 H2O g . If
Calculate the enthalphy of formation for C2H4. DHf CO2 393.5 kJ mol DHf H2O 285.8 kJ mol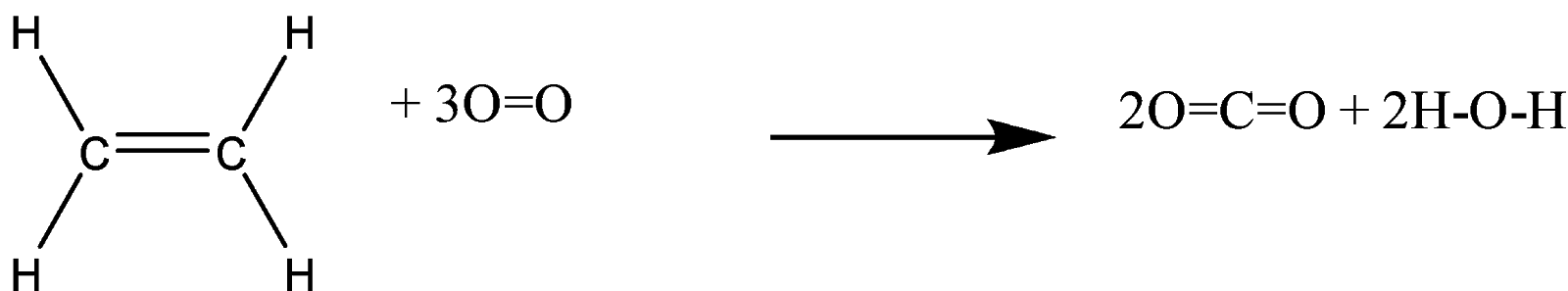
Calculate heat of combustion of ethene from bond energy data C C
Solved 2. The standard enthalpy of combustion of ethylene Chegg
Answered 5. Write a balanced thermochemical bartleby
Calculate the heat of combustion of ethylene to form co and H O
Uncal 39. The heat of combustion of ethene C H. is 1409.3 kJ
The heat of combustion of ethylene at 18 C and at constant volume is 335.8 k
Heats of combustion of C 2 H 4 H 2 and C 2 H 6 are